Translate this page into:
Searching for the Right Evidence: India's Contribution to the Cochrane Pain, Palliative and Supportive Care Group
Address for correspondence: Dr. Hemanshu Prabhakar; E-mail: prabhakaraiims@yahoo.co.in
This is an open access article distributed under the terms of the Creative Commons Attribution-NonCommercial-ShareAlike 3.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms
This article was originally published by Medknow Publications & Media Pvt Ltd and was migrated to Scientific Scholar after the change of Publisher.
Sir,
The Cochrane Collaboration (CC) is spread over 120 countries and is in existence since 1992. The Cochrane Pain, Palliative, and Supportive (PaPaS) Group is one of the 53 Review Groups of the CC. The group publishes reviews of interventions for acute pain arising accidentally or through deliberate injury, chronic pain (lasting 3 months or longer), headache, and migraine, palliative care for those with life-limiting disease or illness and supportive care of patients, and significant others living with serious illness.
We wanted to identify the nationality of review authors in the PaPaS group and highlight the contribution of India in the published work. We collected relevant information from the website of the PaPaS group (www.papas.cochrane.org)*. The retrieved data was entered in to the Excel sheet and analyzed. Data are presented as number (%). A total of 148 review authors were identified for 190 published reviews. The authorship was mainly from the UK [n = 77 (52%)]. This was followed by authorship from the US [n = 13 (9%)] and Australia [n = 10 (7%)] [Figure 1 and Table 1]. India contributed to only 1% authorship. It is evident that most authorship in the PaPaS group is from the UK. There is a need to spread awareness and information regarding the PapaS group in India. There is also a need to encourage authors to contribute to the CC by searching for the right evidence.
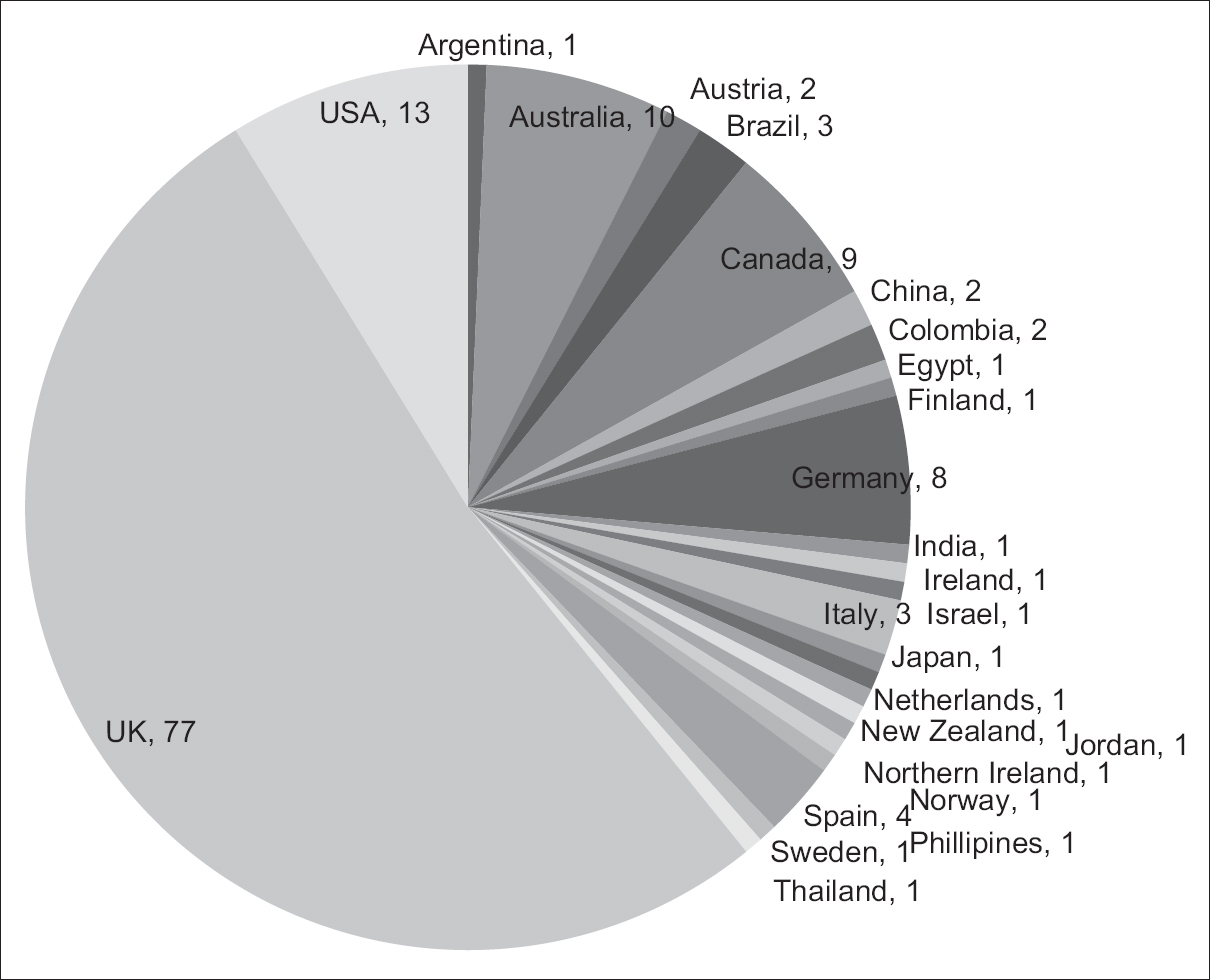
- Representation of review authors from around the world