Translate this page into:
Thyroid Mass: Metastasis from Nasopharyngeal Cancer - An Unusual Presentation
This is an open access article distributed under the terms of the Creative Commons Attribution-NonCommercial-ShareAlike 3.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.
This article was originally published by Medknow Publications & Media Pvt Ltd and was migrated to Scientific Scholar after the change of Publisher.
Abstract
Thyroid gland is an uncommon site of metastasis, and metastasis to the gland secondary to nasopharyngeal carcinoma is seldom seen. We were only able to identify eight reported cases in the literature. A 61-year-old man, diagnosed case of nasopharyngeal cancer–second primary ( first primary-oropharynx), was found to have a thyroid nodule on routine follow-up positron emission tomography-computed tomography (PET-CT) scan. There was no evidence of metastases at any other sites. The thyroid nodule was confirmed as metastatic carcinoma by fine needle aspiration cytology. He was treated with multimodal treatment comprising of surgery followed by reirradiation with concurrent chemotherapy. Subsequently, at the first follow-up (2 months after completion of all treatment), the patient remained asymptomatic, but the response assessment with PET-CT scan was suggestive of lung metastases with no evidence of locoregional disease. Although thyroid parenchymal metastasis is an uncommon occurrence and signifies a poor prognosis, in appropriately selected patients, aggressive therapy with reirradiation and chemotherapy may improve local control and quality of life.
Keywords
Nasopharynx
reirradiation
thyroid metastasis
INTRODUCTION
Nasopharyngeal cancer is an uncommon head and neck cancer (HNC) in most parts of the world. In India, a higher incidence is seen in the northeastern states, particularly Nagaland, Manipur, and Mizoram.[1] It is a chemo- and radio-sensitive tumor and has a high propensity for lymphatic spread and distant metastasis. Radiotherapy (RT) with concurrent chemotherapy is the mainstay in the management of advanced disease.[2]
The incidence of distant metastasis at presentation ranges from 4.4 to 6%. Thirty to 60% of patients with advanced disease develop distant metastasis following treatment.[34] The most common sites of involvement are bone (70%–80%) followed by liver (30%), lungs (18%), and distant lymph nodes (axillary, mediastinal, pelvic, and inguinal). About 98% of them are discovered within 3 years of treatment.[5] Thyroid parenchymal metastasis is an uncommon site of metastasis with an incidence ranging from 0.5% to 24%. The most common primary sites are kidney, lungs, breast, and colorectum with HNCs being a minority (0.02%).[67] We were only able to identify eight reported cases in the literature of intrathyroidal parenchymal metastasis from nasopharyngeal cancer. In this case report, we present a case of nasopharyngeal carcinoma (the second primary in a previously treated case of oropharyngeal carcinoma) with thyroid metastasis managed with surgery and adjuvant reirradiation with concurrent chemotherapy.
CASE REPORT
A 61-year-old male, a resident of Mizoram, presented with complaints of pain while swallowing. Indirect laryngoscopy showed submucosal disease in the left lateral pharyngeal wall in the region of the tonsil. Computed tomography (CT) scan showed heterogeneous-enhancing mass in the left parapharyngeal space measuring 2.4 cm × 2.3 cm × 5 cm with multiple ipsilateral levels II and III lymph nodes [Figure 1a]. Histopathology revealed poorly differentiated squamous cell carcinoma, distinctly different from undifferentiated nasopharyngeal carcinoma. The patient was diagnosed as oropharyngeal carcinoma cT2N2bM0. He was treated with 6 MV photons, using conventional bilateral portals to face and neck with shrinking fields to a dose of 70 Gy/35 fractions with six cycles of concurrent weekly cisplatin. Postradiation clinical examination showed no evidence of disease, and the patient was on regular follow-up.
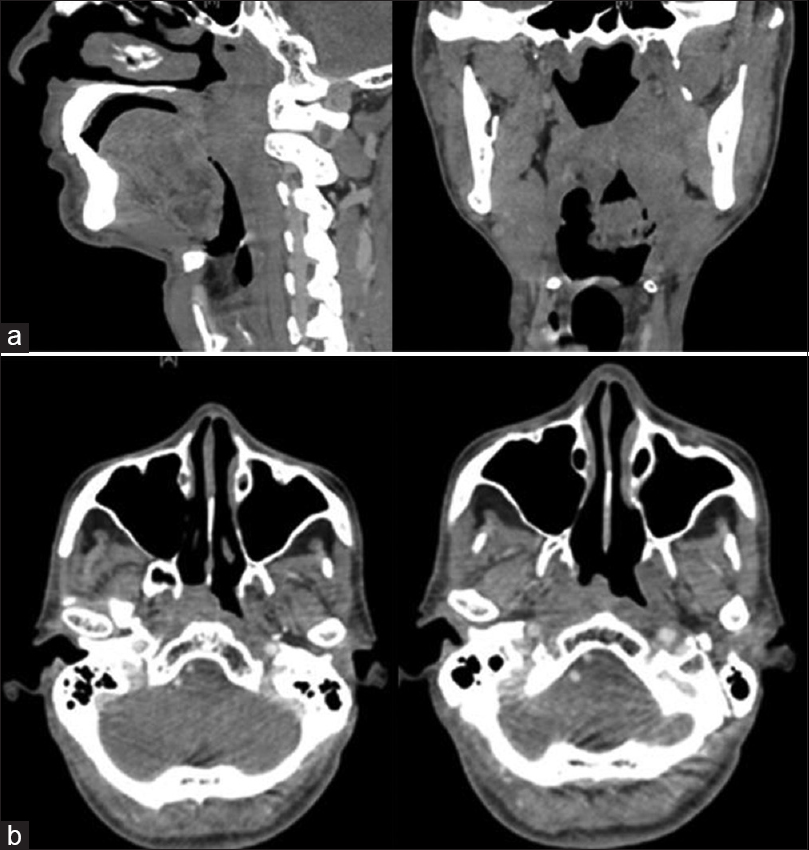
- (a) A sagittal and coronal computed tomography scan showing a left parapharyngeal mass in the region of the tonsil. (b) Axial sections of computed tomography scan showing a soft tissue mass in the right nasopharynx.
After a disease-free interval (DFI) of 5½ years, the patient presented with a swelling in the right axillary region measuring 2 cm × 2 cm. Fine needle aspiration cytology (FNAC) showed poorly differentiated carcinoma. There were no other positive findings on local and systemic examination. A positron emission tomography-CT (PET-CT) was done which showed a localized soft tissue mass in the right nasopharynx, along with a bulky right axillary nodal mass of 4.8 cm × 3.4 cm with no evidence of disease in the oropharynx and neck [Figure 1b]. Biopsy of the nasopharynx was a type IIB, undifferentiated nasopharyngeal carcinoma. The patient was managed as a second primary in the nasopharynx, cT2N0M1 Stage IV. In view of the good general condition, long DFI, and the axillary node being the only site of metastasis, the patient was planned for radical treatment. He received neoadjuvant chemotherapy followed by chemo-RT. He was treated with 9-field intensity-modulated RT (IMRT) with 6 MV photons to face and neck at a dose of 60 Gy in 30 fractions along with six cycles of concurrent weekly cisplatin. The right axillary region was treated with Anteroposterior (AP/PA) portals at a dose of 50 Gy in 25 fractions with 6 MV photons. At follow-up, 3-month posttreatment, PET-CT showed complete response with no uptake anywhere in the body, and nasopharyngoscopy revealed no abnormality.
The patient was on regular follow-up with no complaints. After a DFI of 1 year and 3 months, a routine follow-up PET-CT revealed a 9 mm nodule in the right lobe of thyroid (standardized uptake value [SUV]: 6.73) and a pretracheal node (SUV: 4.53) [Figure 2a and b]. Ultrasonography (USG) of the neck showed hypoechoic lesion in the right lobe of thyroid measuring 1.4 cm × 1.3 cm. A USG-guided FNAC showed poorly differentiated carcinoma cells consistent with metastasis from the nasopharynx [Figure 3]. Since this was the only site of the disease, he underwent right hemithyroidectomy with right levels II–IV neck dissection. Intraoperative findings were suggestive of tumor around the recurrent laryngeal nerve and in the tracheoesophageal groove. A 2 cm × 2 cm node at pretracheal region in superior mediastinum was also excised.
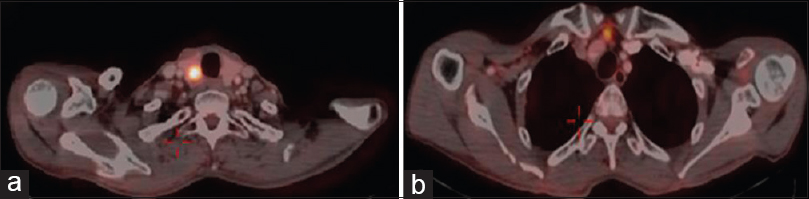
- Positron emission tomography-computed tomography on a routine follow-up showing a nodule in the right lobe of thyroid (a) and a prevascular node (b).
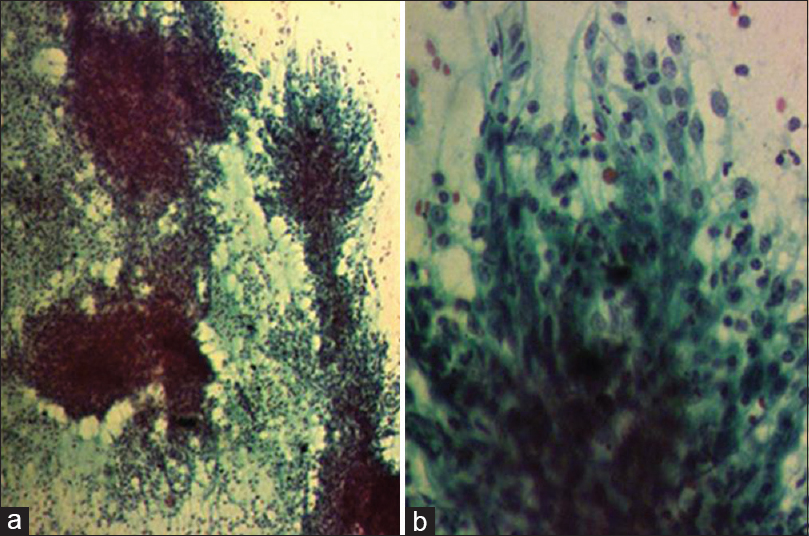
- Cellular smears showing clusters of malignant cells with many lymphocytes in the background. The tumor cells were oval to spindle with scanty cytoplasm, vesicular nuclei, and prominent nucleoli (PAP stain; [a] ×40 and [b] ×200).
Histopathology showed a grayish-white nodule measuring 2.1 cm × 1.5 cm × 1.5 cm located in the right lobe of thyroid abutting the inked margin. Serial sections showed a focus of metastasis of undifferentiated carcinoma, nasopharyngeal type with a heavy lymphoplasmacytic infiltrate. The extrathyroidal extension was noted. The inked margin was focally involved by the tumor. There was no evidence of perineural invasion or lymphovascular emboli. The tumor cells were immunopositive for high-molecular weight cytokeratin and revealed moderate-to-strong diffuse nuclear positivity for Epstein-Barr-encoded RNA/in situ hybridization. The cells were negative for TTF-1, CD23, CEA, and p16. The nodule medial to the recurrent laryngeal nerve showed metastatic deposits of carcinoma. All the nodes in level II–IV were reactive. Both pretracheal nodes showed metastatic carcinoma with perinodal extension [Figure 4a–d].
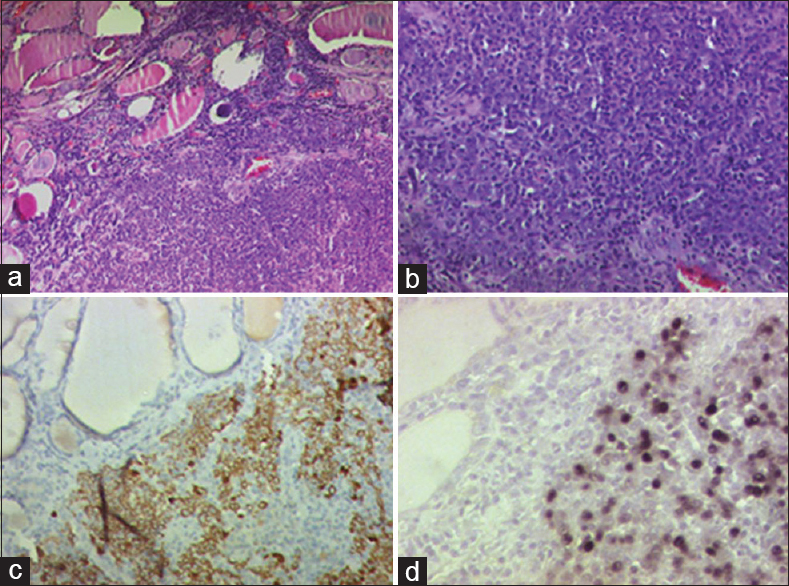
- (a-d) Photomicrograph showing metastatic deposits of undifferentiated nasopharyngeal carcinoma in thyroid with interspersed lymphoid cells (a and b; H and E, ×100 and × 200). The tumor cells were immunopositive for high molecular weight cytokeratin (c; DAB: ×100) and revealed moderate-to-strong diffuse nuclear positivity for Epstein-Barr-encoded RNA/in situ hybridization (d; DAB; ×100).
Although the patient had received radical RT on two previous occasions, the lower neck had received only the elective dose of 50 Gy, and it was felt that the tumor bed and levels VI and VII were at a high risk of recurrence in the presence of positive margins. Hence, he underwent a course of postoperative chemoradiotherapy. He was counseled about the risks of reirradiation and consented to the same. He received a total dose of 60 Gy in 30 fractions to a small CT-planned volume including the thyroid bed, adjacent esophagus, and central compartment of neck including the superior mediastinum (levels VI and VII). Treatment was delivered by a 9-field IMRT plan with 6 MV photons along with concurrent weekly nimotuzumab over 42 days with no gaps in treatment [Figure 5a and b]. He tolerated treatment well with grade II reactions. He remained locoregionally controlled but developed multiple lung metastases after DFI of 8 months. He received palliative chemotherapy and has responded well. He had nociceptive pain over the anterior aspect of the neck associated with dysphagia and odynophagia. He was referred to palliative care services for pain and symptom management. Achieving good locoregional control and palliative care interventions improved his pain, symptoms, and quality of life (QOL) for many months.
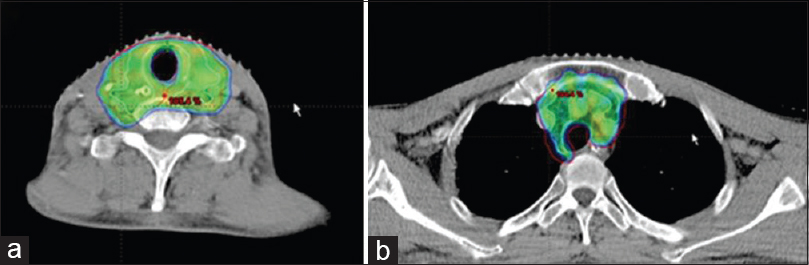
- Axial slice of the planning computed tomography cuts showing adequate coverage of tumor bed (a) and pretracheal node in superior mediastinum (b).
DISCUSSION
HNCs have a high propensity for local recurrence and second primary cancers (SPCs) in view of large area of mucosa exposed to carcinogens. The common sites of elevated risk of SPC are other sites within the upper aerodigestive tract. SPCs are seen in 7.5%–10% of oropharyngeal cancers after definitive treatment.[89] A second HNC is the most common SPC associated with oropharynx. With a better understanding of the biology of cancers and therapeutic advances leading to improved survival, the incidence of SPC has increased.[10] Patients with metachronous SPC from early-stage HNC have a better survival with curative treatment. Presentation of nasopharynx as SPC in oropharyngeal carcinoma is very uncommon, about 0.01% of all cases.[9] In this case, given the long time interval between the two cancers, good performance status of the patient, and the radiosensitivity of the SPC tumor, this patient was considered for reirradiation.
Intrathyroidal metastasis is an uncommon presentation in the clinic though higher incidence rates have been reported in autopsy series (1.25%–24.4%).[11] The most common primary site identified is the kidney.[6] The thyroid is a highly vascular organ in the body being second only to the adrenal gland. The mechanism of metastasis to the thyroid is by direct invasion from adjacent structures, lymphatic spread, or by hematogenous route. Thyroid metastasis from nasopharynx has been reported in only eight cases in literature.[1213141516]
Thyroid metastases rarely cause symptoms. They may present as part of a widely disseminated disease process or may be the first manifestation of metastasis.[17] The usual presentation is a solitary thyroid nodule and can cause symptoms of esophageal or tracheal compression with massive infiltration of the thyroid. Thyroid dysfunction in the form of hypo- or hyper-thyroidism is uncommon. The differential diagnoses include multinodular goiter, primary thyroid cancer such as anaplastic cancer, benign thyroid nodule, follicular dendritic cell tumor, poorly differentiated squamous carcinoma of thyroid, carcinoma showing thymus-like differentiation, and undifferentiated carcinoma. However, in a patient with a history of cancer, the nodule is most often malignant.[18] FNAC is the most common method used to confirm the diagnosis. It is easy, cost-effective, and has high sensitivity, specificity, and negative predictive value.[1920]
Treatment is usually determined by the extent of disease (localized or as a part of disseminated disease) and the DFI between treatment of the index cancer and the metastases. In the absence of any other site of metastases, surgery in the form of lobectomy, hemithyroidectomy, or total thyroidectomy may be carried out.
Our patient underwent hemithyroidectomy as it was a well-defined nodule confined to one lobe of thyroid, with no evidence of distant metastasis. Thyroidectomy, when the thyroid is the only site of metastasis, could help avoid further dissemination of cancer, without affecting the survival.[21] However, when complete resection is not possible due to infiltration of adjacent structures such as trachea and esophagus, there is a high risk of local recurrence which can be very difficult to treat and can cause significant distress to the patient.
The role of adjuvant therapy after distant metastatectomy is debatable. In this scenario, patients may not be offered adjuvant therapy. However, certain factors including the DFI, performance status of the patient, volume and location of disease, and feasibility of delivering effective, salvage treatment determine the utility and efficacy of adjuvant therapy. Two similar cases of thyroid metastasis from nasopharynx have been treated with postoperative RT. Dhouib et al. treated a 50-year-old patient of nasopharyngeal cancer with chemoradiotherapy. Four years later, the patient developed a thyroid mass which was treated with total thyroidectomy followed by concurrent chemoradiotherapy.[16] The second case by Jankowska et al. concerned a 33-year-old woman with recurrent nasopharyngeal carcinoma with thyroid metastasis after an interval of 4 years, who underwent lobectomy followed by postoperative chemoradiotherapy.[15]
In case of primary differentiated thyroid cancer, postoperative external beam RT is indicated in the cases of gross extrathyroidal extension, positive margins, T4-disease, and high nodal burden.[22] In other head and neck squamous cell cancers, patients at a high risk of local recurrence with microscopically involved resection margins or extracapsular spread of disease experience higher locoregional control and disease-free survival with the use of adjuvant concurrent chemoradiation.[23] In this case, the patient had high-risk features in the form of gross residual and microscopic positive margins. As the patient was in good general condition with no distant metastasis or comorbidities and since the low neck had received only 50 Gy in the previous treatments, and moreover the present disease being in the thyroid bed and medially, it was decided to offer the patient a full-course reirradiation with concurrent chemotherapy. Although this was the third time that the patient was receiving radiation, the decision was justified as there was no evidence of distant metastasis and local recurrence which would have a poor outcome with an adverse effect on the QOL of the patient.
Reirradiation in recurrent or second primary HNCs is challenging. Several factors should be kept in mind while considering patients for reirradiation: (1) The indication for reirradiation and alternative treatment available, (2) time since previous treatment, (3) volume to be treated and the degree of overlap between the previously irradiated structures, (4) late sequelae of previous treatment, (5) type of radiation delivery – conformal, (6) and patients' performance status and organ dysfunction. Evidence for reirradiation comes from few retrospective and phase I–III trials. Reirradiation in inoperable patients has poor outcomes with survival rates of 13%–20% but as an adjunct to surgery has shown better disease control and survival.[24]
The patterns of nodal recurrence are difficult to predict in a previously treated neck, whether postneck dissection or post-RT.[25] Nonregional lymph node metastases are also noted, attributable to retrograde dissemination due to blockage of lymphatics. This would have implications on prophylactic lymph node reirradiation. Unexpected lymph node metastasis could occur despite the inclusion of standard first echelon lymph nodes in the target volume. Hence, only the tumor bed and the involved lymph node stations were included in the target volume in our patient.
Conformal techniques such as three-dimensional conformal radiotherapy and IMRT help deliver higher tumoricidal dose with greater normal tissue sparing and lesser toxicity compared to conventional techniques.[26] This is important as the target volume is in proximity to several critical structures such as spinal cord, esophagus, larynx, and trachea. The dose of repeat radiation is another point of concern with a dose more than 60 Gy having better outcomes, however with increased risk of late toxicity, about 30%–40% in the form of skin breakdown, cartilage necrosis, and esophageal stricture.[27] Hence, while deciding on the use of reirradiation, the benefit should outweigh the risks involved. It may definitely be useful in well-selected patients.
This case report highlights how integration of cancer-directed treatment and palliative care interventions improves symptoms and QOL. Laskar and Lewis in their editorial on QOL in HNC report high symptom burden and poor QOL in advanced HNC patients undergoing cancer-directed treatment.[28] An observational study by Lewis et al. have reported a high incidence of pain and distress among HNC patients undergoing chemoradiotherapy.[29] These views were supported by an observational study by Lal et al.[30] An evidence-based review by Talapatra et al. on palliative RT in advanced HNC has recommended using disease-specific QOL instrument for QOL assessment, symptom control, and psychosocial support during the cancer-directed treatment.[31] An AIIMS study of 100 patients of advanced HNC by Gandhi et al. showed that pain, insomnia, fatigue, and loss of appetite were the common symptoms in HNC patients, and there was 50% reduction in global QOL and emotional functioning in these patients.[32] Hence, an integrated, simultaneous, and shared care model of palliative care delivery is essential for prompt management of symptoms and improvement in QOL throughout the illness trajectory.
CONCLUSION
Nasopharyngeal carcinoma arising as the second primary in a known case of oropharyngeal carcinoma is uncommon. Thyroid metastasis from nasopharyngeal carcinoma is rare. Radical treatment should be considered in patients with a good general condition, in the absence of distant metastasis. Surgery followed by chemoradiotherapy in case of high-risk features improves local control. When necessary, reirradiation is a feasible option with optimal results. Concurrent disease-modifying measures with palliative approach improve QOL and achieve symptom control.
Learning points
-
The second primary is one of the long-term sequelae following treatment in HNC
-
Selection of patients for reirradiation is critical. Patients with a good performance status, long DFI, and a small volume of recurrence may be amenable for reirradiation
-
Concurrent cancer-directed treatment and palliative care measures in advanced HNC patients improve symptoms and QOL.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
REFERENCES
- Chemoradiotherapy versus radiotherapy in patients with advanced nasopharyngeal cancer: Phase III randomized Intergroup study 0099. J Clin Oncol. 1998;16:1310-7.
- [Google Scholar]
- Retrospective analysis of 5037 patients with nasopharyngeal carcinoma treated during 1976-1985: Overall survival and patterns of failure. Int J Radiat Oncol Biol Phys. 1992;23:261-70.
- [Google Scholar]
- Clinical outcomes and patterns of failure after intensity-modulated radiotherapy for nasopharyngeal carcinoma. Int J Radiat Oncol Biol Phys. 2011;79:420-8.
- [Google Scholar]
- Distant metastases of nasopharyngeal carcinoma: A study of 256 male patients. J Surg Oncol. 1986;33:194-7.
- [Google Scholar]
- Metastatic tumors of the thyroid gland: A study of 79 cases in Chinese patients. Arch Pathol Lab Med. 1998;122:37-41.
- [Google Scholar]
- Patterns of distant failure and second primary cancers in patients with oropharyngeal squamous cell carcinoma: Implications for surveillance methodology. J Cancer Res Updates. 2014;3:151-61.
- [Google Scholar]
- HPV status and second primary tumours in oropharyngeal squamous cell carcinoma. J Otolaryngol Head Neck Surg. 2013;42:36.
- [Google Scholar]
- Incidence and pattern of second primary malignancies in patients with index oropharyngeal cancers versus index nonoropharyngeal head and neck cancers. Cancer. 2013;119:2593-601.
- [Google Scholar]
- Management and prognosis of metastases to the thyroid gland. J Am Coll Surg. 2005;200:203-7.
- [Google Scholar]
- Thyroid metastasis from nasopharyngeal carcinoma: A case report. J Laryngol Otol. 1994;108:886-8.
- [Google Scholar]
- Detection of thyroid metastases from nasopharyngeal carcinoma with F-18 FDG PET/CT. Clin Nucl Med. 2008;33:224-5.
- [Google Scholar]
- Case report. Isolated intrathyroid metastasis from undifferentiated and squamous carcinoma of the head and neck: The case for surgery and re-irradiation. Br J Radiol. 2008;81:e154-61.
- [Google Scholar]
- A case of thyroid metastasis of an unusual cancer. Cancer Radiother. 2009;13:213-5.
- [Google Scholar]
- Metastases to the thyroid gland: The Royal Marsden experience. Eur J Surg Oncol. 2004;30:583-8.
- [Google Scholar]
- Diagnostic efficacy and importance of fine-needle aspiration cytology of thyroid nodules. J Cytol. 2014;31:73-8.
- [Google Scholar]
- Fine-needle aspiration in the diagnosis of thyroid diseases: An appraisal in our institution. ISRN Pathology 2012:Article ID 912728:4 pages.
- [Google Scholar]
- Metastases to the thyroid gland: Prevalence, clinicopathological aspects and prognosis: A 10-year experience. Clin Endocrinol (Oxf). 2007;66:565-71.
- [Google Scholar]
- Role of adjuvant postoperative external beam radiotherapy for well differentiated thyroid cancer. Radiat Oncol J. 2013;31:162-70.
- [Google Scholar]
- Long-term follow-up of the RTOG 9501/intergroup phase III trial: Postoperative concurrent radiation therapy and chemotherapy in high-risk squamous cell carcinoma of the head and neck. Int J Radiat Oncol Biol Phys. 2012;84:1198-205.
- [Google Scholar]
- Reirradiation for head-and-neck cancer: Delicate balance between effectiveness and toxicity. Int J Radiat Oncol Biol Phys. 2011;81:e111-8.
- [Google Scholar]
- The pattern of failure after reirradiation of recurrent squamous cell head and neck cancer: Implications for defining the targets. Int J Radiat Oncol Biol Phys. 2009;74:1342-7.
- [Google Scholar]
- IMRT reirradiation of head and neck cancer-disease control and morbidity outcomes. Int J Radiat Oncol Biol Phys. 2009;73:399-409.
- [Google Scholar]
- Randomized trial of postoperative reirradiation combined with chemotherapy after salvage surgery compared with salvage surgery alone in head and neck carcinoma. J Clin Oncol. 2008;26:5518-23.
- [Google Scholar]
- Is quality of life “the heart of the issue” in head and neck cancers? Indian J Palliat Care. 2014;20:169-70.
- [Google Scholar]
- Distress screening using distress thermometer in head and neck cancer patients undergoing radiotherapy and evaluation of causal factors predicting occurrence of distress. Indian J Palliat Care. 2013;19:88-92.
- [Google Scholar]
- Initial experience of head and neck cancer patients treated in an oncologist led palliative cancer care clinic at a tertiary cancer care center in Uttar Pradesh: Is the initiative of a full-fledged palliative care for cancer patients justified. Indian J Palliat Care. 2016;22:477-84.
- [Google Scholar]
- Palliative radiotherapy in head and neck cancers: Evidence based review. Indian J Palliat Care. 2006;12:44-50.
- [Google Scholar]
- Symptom burden and quality of life in advanced head and neck cancer patients: AIIMS study of 100 patients. Indian J Palliat Care. 2014;20:189-93.
- [Google Scholar]






