Translate this page into:
Ziconotide: Indications, Adverse Effects, and Limitations in Managing Refractory Chronic Pain
This is an open access article distributed under the terms of the Creative Commons Attribution-NonCommercial-ShareAlike 3.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.
This article was originally published by Medknow Publications & Media Pvt Ltd and was migrated to Scientific Scholar after the change of Publisher.
Sir,
Ziconotide is an N-type calcium channel antagonist which is used to treat chronic pain. It is a synthetic ω-conotoxin MVIIA, a hydrophilic conopeptide toxin which was originally isolated from Conus magus, a species of piscivorous cone snail [Figure 1].[1] At present, ziconotide is the only intrathecal (IT), United States Food and Drug Administration-approved, nonopioid analgesic. Preservative-free morphine is the other drug which is approved for IT use in chronic pain due to cancer and noncancer etiology. IT hydromorphone, clonidine, and baclofen are other drugs used off-label by clinicians. IT baclofen relieves pain due to spasticity in chronic pain conditions. When administered systemically, ziconotide has limited penetration across blood–brain barrier at the cost of unbearable side effects. Therefore, it is administered intrathecally to patients for better penetration across blood–brain barrier, better analgesia, and lesser side effects. Ziconotide blocks N-type calcium channels in the central nervous system, thereby effectively inhibiting neurotransmitter release (norepinephrine) that is responsible for pain at multiple synapses in the nervous system. It also antagonizes N-type calcium channels at the cerebral cortex and the neurohypophysis.[2] IT ziconotide is initially started at a rate of 0.1 μg/h, i.e., 2.4 μg/day as a continuous infusion and is carefully monitored and titrated. The maximum recommended dose is 0.8 μg/h, i.e., 19.2 ug/day. Clinical trials in patients with refractory neuropathic and nociceptive pain have established the feasibility and usefulness of single dose or continuous infusion of IT ziconotide.[3] A continuous IT infusion is managed using an intrathecal drug delivery system (IDSS) infusion or as a patient-controlled analgesia system by the patient. The route of administration of this drug is the hurdle in its regular use. The drug needs to be delivered through an infusion pump (IDSS) and the dose needs to be carefully titrated. Second, adverse events are frequently noted with its use which interrupts the ongoing treatment. The commonly encountered adverse effects are cognitive and neuropsychiatric (confusion, memory impairment, and hallucinations), presyncopal and syncopal episodes, gait disturbances, meningitis, depression, and elevated creatine kinase. Hayek et al. presented their data of 15 patients who were enrolled in ziconotide trial and were followed up for 24 months.[4] Only 4 patients could continue IT medication for 24 months. Rest of the patients had to discontinue it due to the adverse effects. Although the number of cases were less, the cases were followed up for a significant duration to address the adverse effect profile. Recently, Brookes et al. performed a systematic review of randomized controlled trials to investigate the efficacy of ziconotide monotherapy in chronic neuropathic pain.[5] They found that although the monotherapy was effective in pain management, there were serious adverse events which interrupted the ongoing treatment. Ziconotide appears to be a reasonable option for patients with unbearable chronic pain refractory to IT morphine, systemic high-dose opioids, antidepressants, and anti convulsants. The 2016 Polyanalgesic Consensus Conference recommendations are to use ziconotide as an IT monotherapy.[6] However, preservative-free IT morphine or clonidine could be used in refractory patients by careful titration.
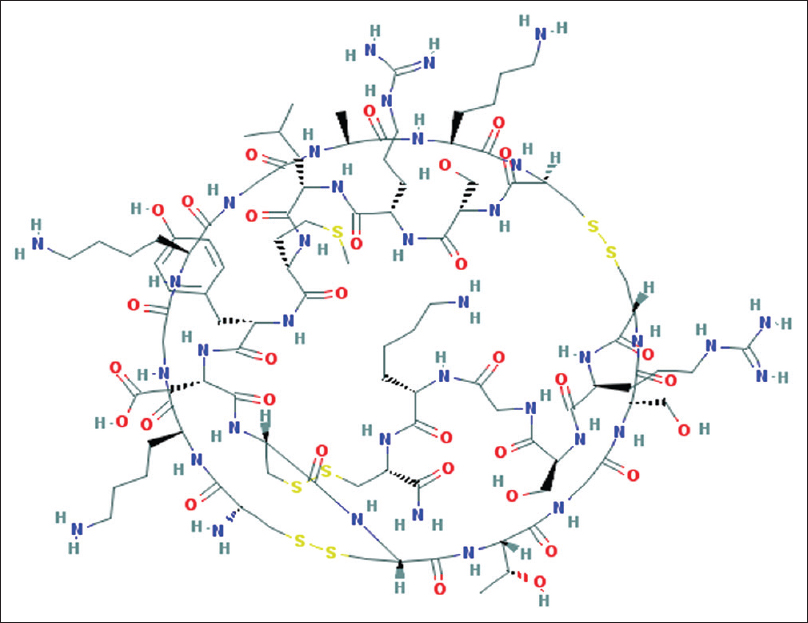
- Chemical structure of Ziconotide. Image source: National Center for Biotechnology Information. PubChem Compound Database; CID = 16135415, https://pubchem.ncbi.nlm.nih.gov/compound/16135415 (accessed June 29, 2017)
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
REFERENCES
- Ziconotide: A review of its pharmacology and use in the treatment of pain. Neuropsychiatr Dis Treat. 2007;3:69-85.
- [Google Scholar]
- Ziconotide: A clinical update and pharmacologic review. Expert Opin Pharmacother. 2013;14:957-66.
- [Google Scholar]
- Intrathecal ziconotide: Dosing and administration strategies in patients with refractory chronic pain. Neuromodulation. 2016;19:522-32.
- [Google Scholar]
- Ziconotide combination intrathecal therapy for noncancer pain is limited secondary to delayed adverse effects: A Case series with a 24-month follow-up. Neuromodulation. 2015;18:397-403.
- [Google Scholar]
- Ziconotide monotherapy: A Systematic review of randomised controlled trials. Curr Neuropharmacol. 2017;15:217-31.
- [Google Scholar]
- The Polyanalgesic Consensus Conference (PACC): Recommendations on intrathecal drug infusion systems best practices and guidelines. Neuromodulation. 2017;20:96-132.
- [Google Scholar]





