Translate this page into:
Anaplastic Carcinoma of the Pancreas: Is There a Role for Palliative Surgical Procedure?
This is an open-access article distributed under the terms of the Creative Commons Attribution-Noncommercial-Share Alike 3.0 Unported, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
This article was originally published by Medknow Publications & Media Pvt Ltd and was migrated to Scientific Scholar after the change of Publisher.
Abstract
Anaplastic carcinoma (AC) or undifferentiated carcinoma of the pancreas is a rare variant among the malignant pancreatic neoplasms. These tumors have a poor prognosis with survival measured in months. The role of surgical palliation to improve the quality of life is not well defined in these patients. We report a case of AC of pancreas in a 65-year-old male patient. Patient had upper abdominal pain with frequent bilious vomiting. Computed tomography scan of the abdomen showed a mass in the body of pancreas with possible infiltration of duodenojejunal flexure (DJF). Laparotomy revealed an inoperable mass with posterior fixity and involvement of the DJF. Patient underwent a palliative duodenojejunostomy. Tissue biopsy from the tumor showed pleomorphic type AC with giant cells. Patient had good symptomatic relief from profuse vomiting and progressed well at follow up. AC of pancreas is a rare and aggressive malignancy with dismal outlook. If obstructive symptoms are present due to duodenal involvement, a palliative bypass may be a worthwhile surgical option in selected cases.
Keywords
Anaplastic carcinoma
Obstruction
Pancreas
INTRODUCTION
Pancreatic carcinoma arising from body and tail of pancreas is uncommon.[1] Anaplastic carcinoma (AC) is a rare (2-5% of all pancreatic cancer) and aggressive variant with extremely poor prognosis. The survival is only for few months from the time of diagnosis.[2] In one study of 35 patients diagnosed with AC of the pancreas, 29 patients died with an average survival of 5.2 months.[3] Their origin is possibly from the ductal cells, as foci of adenocarcinoma have sometimes been noticed in this type of tumor.[4] Most of the reports detail pathological or immuno-histochemical aspects and very few studies describe clinical data and possible treatment options, including palliative procedures, for this aggressive tumor.
We present a case of AC of the pancreas (body/tail region) causing obstruction of the duodenojejunal flexure (DJF). A duodenojejunostomy (DJ) was done for palliation of intractable bilious vomiting.
CASE REPORT
A 65-year-old male patient presented to our hospital with complaints of abdominal pain and bilious vomiting for 4 weeks. Abdominal pain was present in the left upper quadrant, radiating to the back. Vomiting was bilious, frequent, and profuse. Patient had no history of melena, jaundice, or any significant weight loss. Physical examination revealed an ill-defined, firm mass in the left hypochondrium. Blood investigations were normal except for hypokalemia from copious vomiting. CECT scan of the abdomen showed a 6 × 6 cm heterogenous mass arising from the body and tail region of the pancreas [Figure 1]. It had well defined borders and loss of fat plane with relation to the DJF, suggesting infiltration by the mass [Figure 2]. Peri-pancreatic nodes were seen. A diagnosis of pancreatic carcinoma with nodal metastasis was made. An exploratory laparotomy revealed a large, hard, and fixed mass arising from the distal body and tail of pancreas with infiltration of the DJF. There were large metastatic nodes at superior mesenteric and celiac axis. In view of these findings, the tumor was deemed inoperable. Patient underwent a palliative DJ (third part of duodenum and jejunum) in view of symptomatic obstruction to the DJF. A biopsy was taken from the mass and sent for histological study. The patient made an uneventful recovery. Histopathology revealed pleomorphic giant cell type of AC [Figure 3]. Postoperative palliative chemotherapy was not offered as the patient declined. Patient was well, tolerating both fluids and pureed diet at the clinic visit, 4 months after surgery. Later, he was lost to follow up.
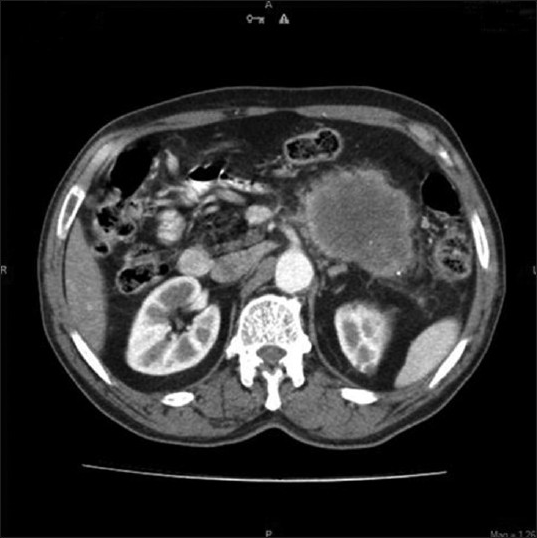
- Large pancreatic mass in body-tail region with peripheral enhancement
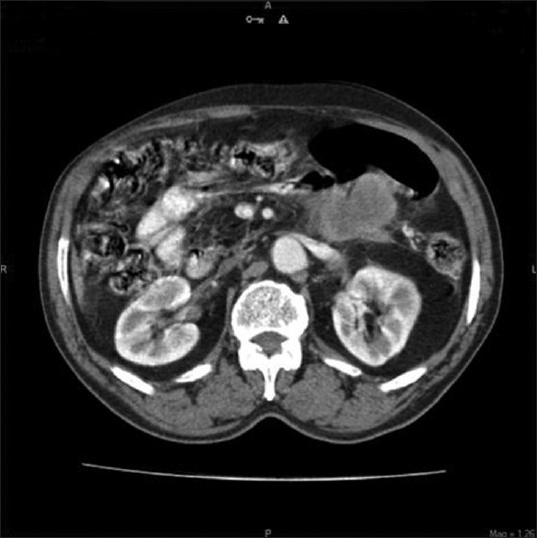
- Pancreatic tumor infiltrating DJ flexure
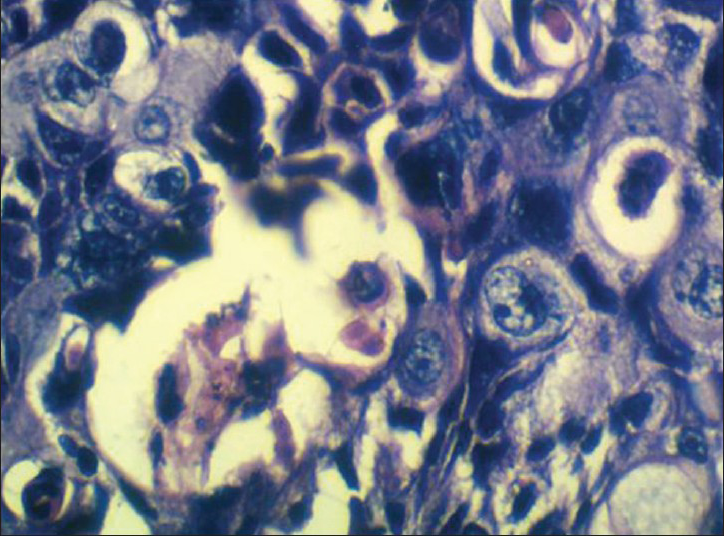
- Histology of tumor showing pleomorphic giant cell pattern
DISCUSSION
Undifferentiated or AC of the pancreas is a rare variant of the exocrine pancreatic malignancies. Three major histopathological variants of AC have been reported in the literature: Spindle cell, pleomorphic, and round cell carcinoma. Among these, the pleomorphic and spindle cell types are frequently displayed in histopathologic examination.[5] The case reported by us had pleomorphic giant cell variant. Prognosis of this type of carcinoma is very poor and has an average survival rate of 5.2 months.[4] Our patient was well till 4 months with good relief from obstructive symptoms and was later lost to follow up. Literature review shows a report with a rare 10-year survival for three patients with other variants of AC.[6]
Establishing a preoperative diagnosis of AC is difficult even with the widespread availability of advanced imaging techniques. This was true in our case, since the features in the abdominal CECT were not helpful in predicting the possible pathological diagnosis. Moore et al., in their report of five patients with pancreatic tumor, had made a preoperative diagnosis of AC with the combination of endoscopic ultrasonogram and fine needle aspiration. The differentiating feature from the typical pancreatic adenocarcinoma was the presence of well demarcated hyper and hypoechogenic areas in the suspected lesion, located close to each other.[78]
Possible reasons for aggressive nature of this malignancy are loss or impaired expression of surface adhesion molecules like E-cadherin, alpha and beta catenin.[9] Due to rapid downhill course, treatment options including palliative procedures are very much limited. There is no evidence-based data available to support any particular intervention. Few studies have compared RO/R1 resection with palliative surgery for these tumors. It was found that median survival was greater for resective procedures and for neoplasms containing osteoclast like giant cells.[10] No reports are available in the literature regarding DJF obstruction due to these tumors and the role for any palliative bypass procedure. This palliative option should always be considered in inoperable cases with symptomatic DJF obstruction, with an aim to provide reasonable quality of life for the remaining survival period.
Clinical response of this particular variant of pancreatic cancer to available adjuvant therapy is usually poor. Recent studies have shown expression of L1CAM, COX-2, and EGFR in majority of the AC and these may potentially become putative therapeutic targets.[11]
CONCLUSION
AC is a rare malignancy with dismal prognosis and median survival measured in months. In spite of these discouraging features, a palliative bypass may be a worthwhile option, in selected patients, for alleviating troublesome obstructive symptoms.
Source of Support: Nil
Conflict of Interest: None declared.
REFERENCES
- Anaplastic carcinoma associated with a mucinous cystic neoplasm of the pancreas during pregnancy: Report of a case and a review of the literature. World J Gastroenterol. 2008;14:132-5.
- [Google Scholar]
- A clinicopathologic and immunohistochemical study of 35 anaplastic carcinomas of the pancreas with a review of the literature. Ann Diagn Pathol. 2001;5:129-40.
- [Google Scholar]
- Pancreas and Periampullary region. In: Rosai J, ed. Ackerman's Surgical Pathology. Philadelphia: Mosby – Year Book; 1996. p. :981-2.
- [Google Scholar]
- Anaplastic carcinoma of the pancreas associated with a mucinous cystic adenocarcinoma. A case report and review of the literature. JOP. 2007;8:775-82.
- [Google Scholar]
- Anaplastic ductal carcinoma of the pancreas with more than 10 year postoperative survival: Report of 3 cases. Pancreas. 2006;33:88-9.
- [Google Scholar]
- Osteoclastic and pleomorphic giant cell tumors of the pancreas diagnosed via EUS-guided FNA: Unique clinical, endoscopic, and pathologic findings in a series of 5 patients. Gastrointest Endosc. 2009;69:162-6.
- [Google Scholar]
- Osteoclastic and pleomorphic giant cell tumors of the pancreas: A review of clinical, endoscopic, and pathologic features. World J Gastrointest Endosc. 2010;2:15-9.
- [Google Scholar]
- Phenotypical characteristics of undifferentiated carcinoma of the pancreas: A comparison with pancreatic ductal ade-nocarcinoma and relevance of E-cadherin, alpha catenin and beta catenin expression. Oncol Rep. 2001;8:745-52.
- [Google Scholar]
- Anaplastic pancreatic carcinoma: Presentation, surgical management, and outcome. Surgery. 2011;149:200-8.
- [Google Scholar]
- Expression of L1CAM, COX-2, EGFR, c-KIT and Her2/neu in anaplastic pancreatic cancer: Putative therapeutic targets? Histopathology. 2010;56:440-8.
- [Google Scholar]






