Translate this page into:
Assessment of Anxiety and Depression in Oral Mucositis Patients Undergoing Cancer Chemoradiotherapy: A Randomized Cross-sectional Study
Address for correspondence: Dr. Danam Reshma Priyanka; E-mail: drreshmapriyanka@gmail.com
This is an open access article distributed under the terms of the Creative Commons Attribution-NonCommercial-ShareAlike 3.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.
This article was originally published by Medknow Publications & Media Pvt Ltd and was migrated to Scientific Scholar after the change of Publisher.
Abstract
Introduction:
Oral mucositis is an unavoidable complication occurring during the treatment of cancer by radiotherapy, concurrent chemoradiotherapy (CCRT), or chemotherapy. This is a painful sequelae, significantly affecting the nutritional intake and quality of life.
Materials and Methods:
A multicentric cross-sectional study was done at four cancer centers in Hyderabad. About 455 subjects of both genders between 20 and 80 years undergoing cancer treatment such as chemotherapy (Group I), CCRT (Group II), radiotherapy within 14 days of initiation (Group III), and radiotherapy after 14 days of initiation of therapy (Group IV) who had oral mucositis were included in the study. A self-addressed Hospital Anxiety and Depression Scale questionnaire was used to assess the anxiety/depression in cancer therapy-induced oral mucositis patients.
Results:
Group III had established anxiety (58.82%) followed by Group IV (47.5%) of patients showing severe oral mucositis. In Group I, 47.17% and in Group II, 40% patients with borderline anxiety had mild mucositis, which was statistically significant (P = 0.01). Group III had established depression in 56.36%, followed by Group IV with 39.62% patients showed severe oral mucositis. Group I and II had mild to moderate mucositis, which was associated with established depression at statistically significant result (P = 0.02).
Conclusion:
Group IV had maximum participants with anxiety and depression, closely followed by Group II, Group III, and least in Group I. Thus appropriate intervention in the form of nonpharmacological and pharmacological treatment is warranted.
Keywords
Concurrent chemo-radiotherapy
Chemotherapy
Diagnostic and Statistical Manual of Mental Disorders
Hospital Anxiety and Depression Scale
International Classification of Diseases
Radiotherapy
INTRODUCTION
Oral cancer is one of the most common cancer and is the sixth leading cause of mortality.[12] In India, it is the second most common cancer in males following bronchogenic carcinoma. Treatment often involves surgical intervention, radiotherapy, concurrent chemoradiotherapy (CCRT), or chemotherapy alone, depending on the stage of lesion and responsiveness of the patient. Oral mucositis is an unavoidable complication which occurs during the treatment of cancer.[1234] Patients treated with radiation therapy for head and neck cancer receive approximately 200 cGy daily dose of radiation, 5 days/week, for 5–7 continuous weeks. It was reported that severe oral mucositis prevailed in 29–66% of patients receiving radiotherapy.[2] About 75–85% of patients who receive high-dose chemotherapy prior to hematopoietic stem cell transplantation develop oral mucositis.[2]
Oral mucositis can be very painful, significantly affecting the nutritional intake, mouth care, and quality of life.[123] This may prompt the oncologist to defer the treatment without completing the full course of treatment. Apart from oral mucositis, it was reported that the patients suffered from pain, fatigue, xerostomia, dysphagia, sleep disturbances, and cognitive dysfunction, thus negatively impacting the quality of life.[15]
One third of all cancer patients receiving oncological treatment and those with advanced cancers suffer from mental distress to an extent.[4] Physicians unwittingly fail to recognize the distressed patients invariably adding to their already compromised oral function and general well-being.[1] In a cancer context, emotional distress can be conceptualized as referring to anxiety, depression, and adjustment disorders related to the cancer experience.[6] Depression was found to be a persistent component at various stages of cancer diagnosis, treatment, and after.[1] A study reported the prevalence of depression in cancer patients between 25% and 52% during treatment and 11–45% at 6 months follow-up.[1] Prevalence of distress reported as such in palliative care varies between 7% and 60% depending on the stage and type of disease, study design, and measures used, with most studies suggesting that around one-third of patients experience clinically significant distress.[6] Adjustment disorders are the most common psychiatric syndrome seen in cancer patients. Derogates et al. reported its prevalence as 68% in their study population.[7]
Once identified, psychological distress in terminally ill patients is amenable to treatment through good psychological support and medications.[6] Data suggest that screening for and addressing distress not only enhances quality of life but also may be associated with improved cancer outcomes.[8]
Emotional and psychological factors can disturb a wide variety of hormonal, vascular, and muscular functions, all of which may produce peripheral changes varying from pain to oral ulceration. Concomitant oral mucositis and anxiety/depression can modulate response of patient toward pain perception.[9] Thus, need of the hour was to evaluate anxiety/depression levels in patients having oral mucositis so that early recognition can decrease the morbidity associated with cancer treatment.
Rates of distress in cancer are measured as self-reports, with cutoff scores signifying distress or as combined rate of psychological disorders, mainly anxiety, depression, and adjustment disorders measured by psychiatric diagnostic interviews.[6] Various scales have been used to evaluate distress in the individuals with cancer, one of which is the popular Hospital Anxiety and Depression Scale (HADS). HADS – is a brief self-administered scale, which has been specifically designed for use in patients with co-morbid physical illness. It consists of 14 items, seven each recording depression and anxiety. Each item has four possible choices scoring from ′0′ to ′3′. The depression subscale has been constructed in such a way as to largely exclude somatic symptoms. This subscale consists of items that predominantly screen for anhedonia, which is considered a symptom characteristic of the endogenous subtype of depression.[10] Validation studies have established a high internal consistency and reliable factor structure. The scale has been validated in the Indian population in a study that used HADS to screen for depression and anxiety.[11] They also established sensitivity and specificity for various cut-off scores. The study suggested that the best cut-off values for the Indian population are ′8′ and ′7′ for depression and anxiety, respectively. A cut-off score of ′8′ on a depression subscale has a sensitivity of 75% and specificity of 76%, respectively, for the diagnosis of depression.
It is one of the commonly used questionnaire scales for identifying anxiety/depression in oncological settings.[4] The fourteen item questionnaire with cut off points indicates oncologists to decide about including a mental health care professional in treatment planning.[412] There are very few studies relating anxiety and depression among oral mucositis patients on large scale population in Indian context. The present self-addressed HADS questionnaire study was intended to assess the anxiety/depression in cancer therapy-induced oral mucositis for appropriate pharmacological or nonpharmacological approach thereby improving the quality of life and elevating the signs and symptoms.
MATERIALS AND METHODS
This multicentric randomized cross-sectional study was conducted at various oncological centers namely MNJ Government Hospital, Yashoda Cancer Institute, Kamineni Institute of Medical Sciences and Bibi Cancer Hospitals of Hyderabad city. A total of 455 subjects of both genders between the age group of 20–80 years who were undergoing cancer treatment in the form of chemotherapy, radiotherapy, and CCRT and suffering from oral mucositis at different stages were included in the study.
The sampling was randomized, and the patients were divided into four groups depending on the treatment they were undergoing as the intention was to compare the severity of oral mucositis which differs with the type of treatment and even with the duration of treatment (so the patients undergoing radiotherapy were divided into two groups depending on duration) along with anxiety and depression, Group I consisted of patients undergoing chemotherapy and having oral mucositis. Group II included those patients undergoing CCRT and having oral mucositis. Group III included radiotherapy patients within 14 days of initiation of therapy and having oral mucositis. Group IV included radiotherapy patients after 14 days of initiation of therapy and having oral mucositis.
Inclusion criteria were diagnosis of cancer, ability to speak and to complete the questionnaire, and willingness to be part of the study. The study was approved by the Local Ethics Committee.
HADS scale was translated into regional language and validated. This allowed us to attain uniformity among the questionnaire participants and reducing the discrepancy of scores obtained during the study. The present study was carried out over 4 months period from June 2015 to September 2015. Participants were selected from outpatient and inpatient hospital facilities in the abovementioned oncological centers. The inpatients mostly fell into chemotherapy group and CCRT groups. The questionnaire was distributed among all the participants, and it was immediately collected once answered. The questionnaire had seven questions pertaining to anxiety and seven questions pertaining to depression. The summed up scores for anxiety and depression separately were obtained with the range between 0 and 7 considered as normal, 8–10 considered as borderline, and 11 and above were considered as being abnormal or having frank anxiety/depression. The scores thus obtained were subjected to final assessment under six different variables among all 455 participants.
In addition, the severity of oral mucositis was also evaluated in all 455 patients grouping them under mild, moderate, and severe. The treatment of pain and burning from oral mucositis was done under the supervision of concerned oncologist who was in charge of the patient at presentation. The data regarding such treatment were not shared with the study investigators due to confidentiality concerns pointed out by the oncologist regarding the patient treatment protocols from these four hospitals.
The data, thus generated was statistically analyzed using Chi-square and ANOVA test analysis with P value significant at <0.05.
RESULTS
Gender
A total of 455 participants took part in the study. Among them, 268 (58.90%) were females and 187 (41.10%) were males with significant P value, P = 0.0011 [Table 1].

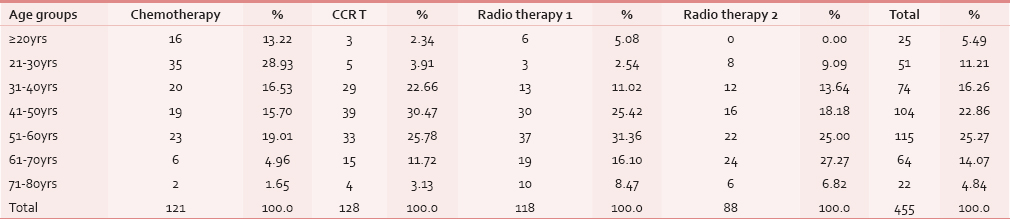
Age
The participants’ age ranged from 20 to 80 years. Maximum number of people participated in the survey belonged to the age group between 51 and 60 years, 115 (25.27%) and least between 71 and 80 years, 22 (4.84%), with P value significant P = 0.000001. Considering separate groups, there were no participants below 20 years in Group IV. Maximum number of participants 30.47% were in Group II between the age group 41–50 years, and least with 1.65% were in Group I between the age group 71–80 years. When one-way ANOVA was applied for comparison of four groups with mean age, a statistically significant P value was obtained (P = 0.00001) [Table 2].
Duration of treatment
Group I was not considered here as there was a wide variation in the treatment duration of chemotherapy group participants. Hence, comparison of remaining three groups with mean duration of treatment by one-way ANOVA was done, and it yielded statistically significant P value (P = 0.00001) in all the groups.
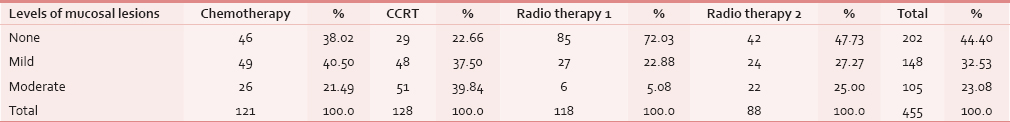
Intensity of mucosal lesions/oral mucositis
The oral cavity was examined and the oral mucositis in all the groups were divided into three subgroups based on intensity of lesion as mild, moderate, and severe. Out of 455 patients, 202 (44.4%) presented with severe lesions, 148 (32.53%) presented with mild lesions, and 105 (23.08%) presented with moderate lesions with P value statistically significant (P < 0.05). When individual groups were assessed, severe mucositis was present in Group III followed by Group I, Group IV, and least in Group II. Mild mucositis was found maximum in Group I and least in Group IV, moderate mucositis was found maximum in Group II, and least in Group III [Table 3].
Diet
The diet intake was divided into solid, liquid, and no intake possible. The groups where compared with intake of the form of diet. Out of 455 participants, only liquid diet intake was found in 106 (23.30%) and solid intake was in 349 (76.70%). There were no participants under “oral alimentation impossible” category. P value was significant among all four groups and closer significance was found in Group I versus Group IV.
Assessment of anxiety
Anxiety levels were divided into normal, borderline, and abnormal and comparisons were drawn in all the four groups with all levels of anxiety.
Out of 455 participants, 181 (39.78%) participants had borderline anxiety, which was highest when compared with other levels of anxiety, followed by 149 (32.75%) participants having abnormal anxiety and 125 (27.47%) participants had no anxiety. When comparison of four groups with levels of anxiety was carried out, there was no statistically significant P value among four groups. There were comparable anxiety levels in Group I versus Group IV and Group III versus Group IV with significant P value (P < 0.05) [Table 4].

Assessment of depression
Out of 455 participants, 234 (51.43%) participants had abnormal depression, 140 (30.77%) participants were in borderline stage, and 81 (17.8%) participants had no depression. Large number of participants having abnormal depression belonged to Group II 75 (58.59%), followed by Group III 55 (46.61%), Group IV 53 (60.23%), and Group I, 51 (42.15%). Borderline depression was found equally among Group I and Group II with P value statistically significant in all the four groups. Closest significance was found between Group I versus Group II with P = 0.004 [Table 4].
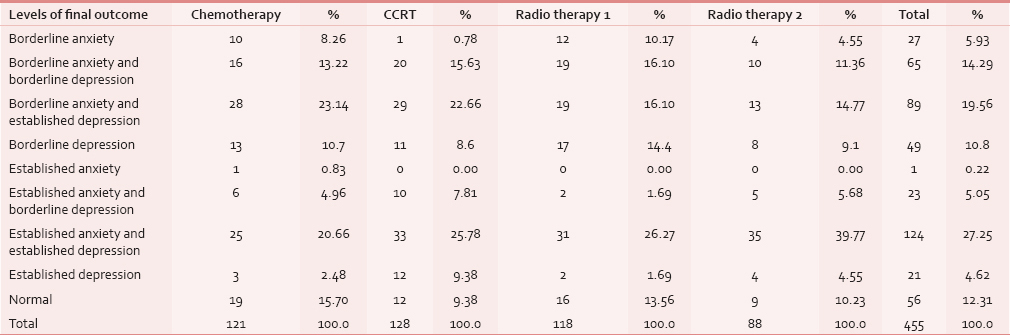
Comparison of four groups with levels of final outcome
Total numbers of six variables were obtained as final outcome, under the category of anxiety and depression.
Out of 455 participants, 124 (27.25%) participants showed established anxiety and established depression which was maximum in the study population followed by 89 (19.56%) participants with borderline anxiety and established depression, 65 (14.29%) participants had borderline anxiety and borderline depression, 49 (10.8%) participants with borderline depression only, 27 (5.93%) participants with borderline anxiety only, 23 (5.05%) participants with established anxiety and borderline depression, and 21 (5.93%) with established depression only. Fifty-six out of 455 participants were not anxious or depressed. The above scores were derived based on HADS.
All the groups except Group I had maximum number of people suffering from established anxiety and established depression and in Group I participants had borderline anxiety and established depression. Out of 21 subjects with established depression only, 12 (9.38%) belonged to Group II, 4 (4.55%) in Group IV, 3 (2.48%) in Group I and 2 (1.69%) in Group III. There was only 1 (0.83%) patient with established anxiety in Group I. There were 56 (12.34%) patients who had no anxiety or no depression of which maximum were seen in Group I 19 (15.7%) [Table 5].
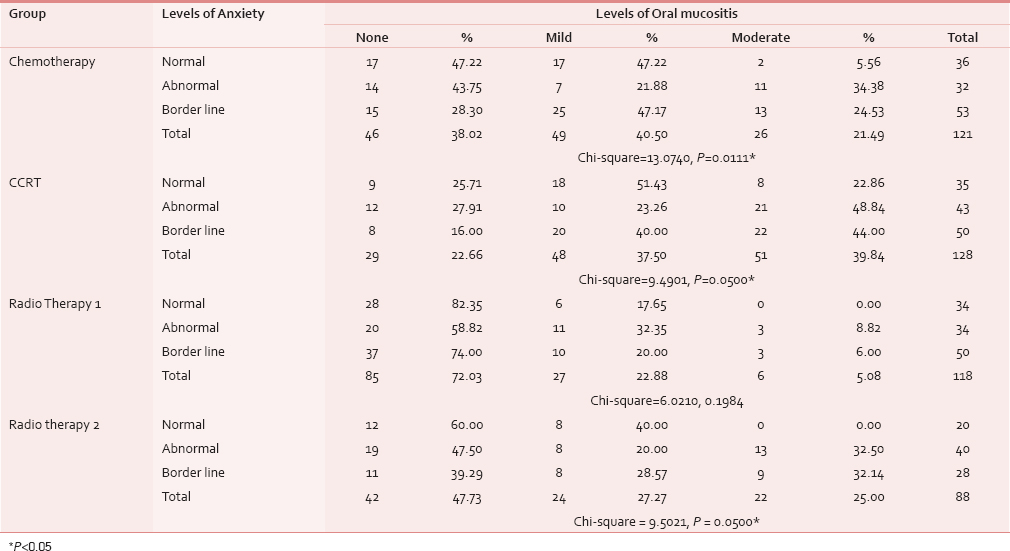
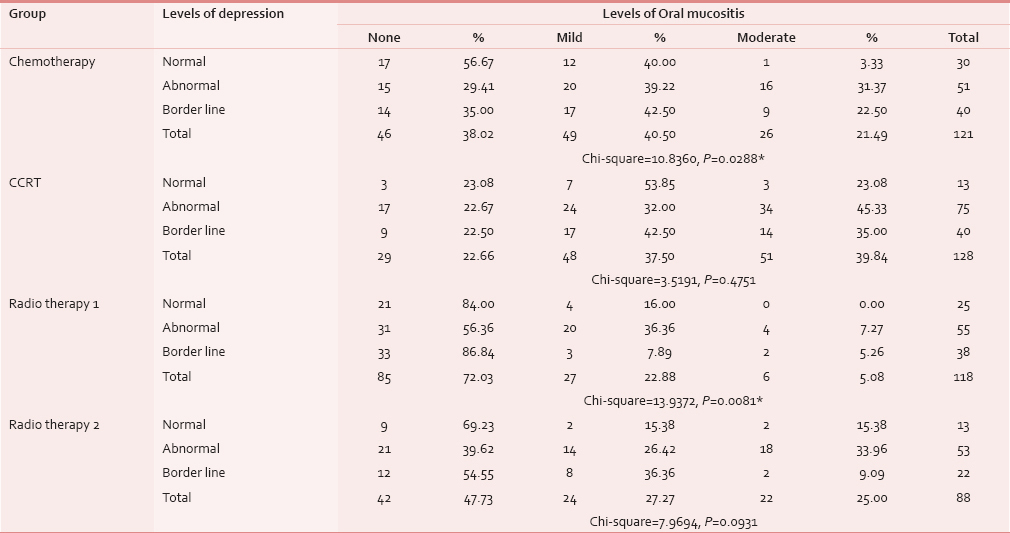
Association of levels of oral mucositis with levels of anxiety
Table 6 shows Group III had established anxiety in 20 (58.82%) out of 34 patients followed by Group IV with 19 (47.5%) of 40 patients showing severe oral mucositis. In Group I, 25 (47.17%) patients and in Group II, 20 (40%) patients with borderline anxiety had mild mucositis, which is statistically significant (P = 0.01).
Association of levels of oral mucositis and levels of depression
Table 7 shows Group III had established depression in 31 (56.36%) out of 55 patients, followed by Group IV with 21 (39.62%) of 53 patients showing severe oral mucositis. In Group I as well as in Group II, mild mucositis was associated with abnormal depression and moderate mucositis was associated with established depression, which is statistically significant (P = 0.02).
DISCUSSION
Numerous clinical studies have focused on mucosal toxicity associated with cancer therapy, which is a common acute toxic effect of radiotherapy as well as CCRT. Severe mucositis can be distressing and may lead to decreased intake of food leading to malnutrition.[3]
Several studies have demonstrated that 30–40% of patients with cancer report emotional distress as a consequence of the disease and treatment, many of whom meet the criteria for adjustment, anxiety, and depressive disorders.[13] Psychological distress, a common yet treatable condition in individuals with cancer, is associated with suffering and poor outcomes.[8] Depressive symptoms can be caused by the disease process directly or by the various chemotherapeutic agents used for the treatment of cancer.[14] Depression is a common psychiatric manifestation occurring during and also after cessation of cancer therapy.[19] Studies have evaluated the scores related to depression among cancer patients undergoing treatment but were not specific during oral mucositis stage.
Adjustment disorders entered the DSM-II nomenclature in 1968 and were recognized in ICD-9 in 1978. They are conceived of as developing in response to a variety of causal stressful events, the symptoms representing an adaptation to these stressors or to their continuing effects.[15] The predictive validity of the diagnosis of adjustment disorder has been confirmed also among adult inpatients (Andreasen and Hoenk, 1982),[16] with 79% of adults being well 5 years after the index admission. In those with adjustment disorder most symptoms resolve rapidly (Snyder et al., 1990;[17] Despland et al., 1995),[18] with fewer than 17% developing a chronic course (Bronisch, 1991;[19] Greenberg et al., 1995).[20]
There is a scarcity of literature on studies associating anxiety and/or depression to oral mucositis during various cancer therapies excluding surgery. The present study is done to evaluate the presence of anxiety and depression in oral mucositis patients undergoing radiation therapy, chemotherapy, or CCRT using HADS scale.
Scale: Numerous screening tools varying in length and complexity are available, but establishing the validity of a screening tool in a specific patient population is vital before it can be recommended for routine use.[6]
More than 200 published studies from most medical settings worldwide have reported experiences with the HADS, which was specifically developed by Zigmond and Snaith in 1983[10] for use with physically ill patients.[21] Its purpose was to provide clinicians with an acceptable, reliable, valid, and easy to use practical tool for identifying and quantifying anxiety and depression. The role described under HADS was a dimensional rather than categorical one; it can be best used not to make diagnosis of psychiatric disorders, but for identifying general hospital patients who need further psychiatric evaluation and assistance.[22]
HADS has been translated and widely used in more than 25 countries since its original development. Herman in an extended review reported that HADS had demonstrated reliability and validity when used to assess medical patients.[22]
A study was conducted on 521 participants to validate the Greek translation of HADS and assess its psychometric properties and concluded that HADS showed good psychometric properties and could serve as a useful tool for clinicians and it presented higher consistency and reliability.[22]
For rapid assessment of psychiatric morbidity in cancer patients, the hospital anxiety and depression scale have been adapted to test Indian cancer patients.[10] The cut off score of 7 on the anxiety subscale gives a sensitivity of 87% and specificity of 79%. For depression subscale, the cutoff score of 8 gives a sensitivity of 75% and specificity of 76%. If one uses the total scores, the cutoff 16 gives a sensitivity of 85% and a specificity of 88%.[14]
The efficacy of screening questionnaires in detecting all forms of psychological disorders (conceptualized as distress) in palliative care patients, most of whom had advanced cancer and the three touch screen-based screening tools (depression thermometer [DT], brief symptom inventory [BSI-18], general health questionnaire-12 [GHQ-12]) had performed equally well in identifying distress compared with a psychiatric interview. DT, a single item visual analog scale, which could be easily completed and interpreted, performed equally well as the GHQ-12 or BSI-18, both of which were lengthier and required some expertise to interpret.[6] DT is a simple and effective screening instrument for detecting distress in Italian patients with cancer.[11]
Although the distress thermometer is widely used in cancer supportive and palliative care across a diverse range of settings and in many populations, there is little evidence to support its validity and little is known about what the tool actually measures.[23]
To make most cost effective screening of mental disorders feasible, HADS-Anxiety and HADS-Depression questionnaire scale have been used in various studies. HADS was found to perform well in assessing severity and caseness of anxiety disorders and depression in both somatic and psychiatric and primary care patients and in general population.[912]
Limitation of Hospital Anxiety and Depression Scale
There is still no comprehensive documentation of its psychometric properties. The HADS gives clinically meaningful results as a psychological screening tool, in clinical group comparisons and in correlational studies with several aspects of disease and quality of life. It is sensitive to changes both during the course of diseases and in response to psychotherapeutic and psychopharmacological intervention. HADS scores predict psychosocial and possibly also physical outcome.[21]
Many questionnaires devised in the West have been standardized to assess the psychological status of cancer patients throughout the world, one of which HADS was used in the study. However, some questions seemed unsuitable for our patients; hence, a questionnaire for the Indian set up is desirable.[24] In our study, many patients faced the problem of understanding idioms such as “butterflies in the stomach” even when translated into local language and between certain options, requiring us to provide the help of principal translator.
The validity of HADS after its translation, when not addressed carefully, the influence of language or culture might manifest causing shift in mean scores or diminished validity or it could lead to incomparable cut points because the translated item measures something else than intended in the original version.[25]
HADS has been used in this study to evaluate the participants taking part in the survey. Four hundred and fifty-five participants agreed to be part of the study, and there were no withdrawals.
Gender
Of the 455 participants, 268 (58.9%) were women and 189 (41.1%) were men. This may represent the gender characteristics of cancers in general in this region of India. Maximum numbers of female participants were in Group III with 72.88%. This was found similar to the study “Psychological factors associated with head and neck cancer treatment and survivorship” by Howren et al.[26]
Groups
Ideally, radiotherapy is given for 4–5 weeks, 5 days a week between 100 and 200 cgy depending on the type of cancer. Radiotherapy group was divided into two subgroups-one, within 15 days of initiation of therapy and second group, after 15 days of therapy. This was done to compare the advent of oral mucositis during the second week of radiotherapy and healing during last week of treatment. Chemotherapy group was not divided as the participants who were undergoing therapy for more than a year might have affected the scores in the study. In addition, subjects undergoing CCRT were also included as separate group.
Age
The age of participants ranged from 20 years to 80 years, with the maximum in 51–60 years and least in the age group 71–80. This was found to be similar to a study on Taiwanese oral cancer patients “Exploring Changes in Symptom Severity and Depression during Cancer Treatment.”
Intensity of mucositis versus anxiety and depression
This variable when correlated with presence of anxiety and/or depression demonstrated significant results. It is a well known fact that as the treatment of cancer increased in number of days or cycle, the mucositis intensity increases.[149] Thus, along with the changes in intensity, there was significant increase in the scores of anxiety and depression in all the groups. It was most prominent in Group IV patients. This finding had corroborated with similar other study in breast cancer patients undergoing chemotherapy.[27]
The levels of anxiety when correlated with the levels of mucositis showed that those in Group III had established anxiety closely followed by the Group IV. All the participants had severe mucositis in oral cavity. The appearance of mild mucositis and moderate mucositis was associated with borderline anxiety in Group I and Group II. This is conflicting evidence when compared with depression scores where the Group I and Group II with mild mucositis and moderate mucositis had established depression.
Diet
The dietary intake of solid and liquid food might have been compounding factors in the study. The patients on only liquid diet belonged majorly to Group II, followed closely by Group IV. Majority of participants were on solid food intake in Group I. However, conclusion could not be arrived regarding the scores of anxiety or depression with that of diet alone. There might have been more compounding factors along with the diet which would have influenced the scores.
Not anxious/depression
It was noteworthy that out of 455 participants, just 56 were classified as not anxious/depressed using HADS. Most of such subjects were from Group I.
CONCLUSION
From the above study, it can be concluded that the Group IV had most number of participants with established anxiety and established depression. This was closely followed by Group II, then by Group III and least by Group I.
Thus, appropriate intervention in the form of nonpharmacological and pharmacological treatment appears warranted in all the above groups. Many of the antianxiety and antidepressants themselves cause profound oral changes such as hyposalivation, which may modify the pain, severity, and effecting quality of life. Appropriate psychiatric interventions during the treatment course as a multidisciplinary approach can better help and may improve overall treatment outcome in cancer treatment.
Limitations of the study
Certain limitations marred the definitive outcome of the study which may be overcome in further other research directed toward such a process. There was no follow-up of the patients after 6 months to check for changes in the scores of anxiety or depression. The study also did not look into other compounding factors such as financial background and personal habits, which could have had an influence on the scores of anxiety or depression. The site and type of cancer might have influenced the scores, which again were ignored. An association between socioeconomic status and education, with anxiety and depression were not considered. Staging of the cancer and pain, which would have definitely affected the scores, was not taken into account due to confidential patient records which were not shared with the study investigators. It is recommended to include such factors and evaluate patients at various time intervals to arrive at proper analysis for effective intervention strategy.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
REFERENCES
- Changes of symptoms and depression in oral cavity cancer patients receiving radiation therapy. Oral Oncol. 2010;46:509-13.
- [Google Scholar]
- Management of oral mucositis in patients who have cancer. Dent Clin North Am. 2008;52:61-77.
- [Google Scholar]
- Assessment of cancer therapy-induced oral mucositis using a patient-reported oral mucositis experience questionnaire. PLoS One. 2014;9:e91733.
- [Google Scholar]
- Hospital anxiety and depression scale cutoff scores for cancer patients in acute care. Br J Cancer. 2009;100:908-12.
- [Google Scholar]
- Impact of oral mucositis and its sequelae on quality of life. J Support Oncol. 2004;2:79-80.
- [Google Scholar]
- Screening for psychological distress in palliative care: Performance of touch screen questionnaires compared with semistructured psychiatric interview. J Pain Symptom Manage. 2009;38:597-605.
- [Google Scholar]
- The prevalence of psychiatric disorders among cancer patients. JAMA. 1983;249:751-5.
- [Google Scholar]
- Recommendations for the implementation of distress screening programs in cancer centers: Report from the American Psychosocial Oncology Society (APOS), Association of Oncology Social Work (AOSW), and Oncology Nursing Society (ONS) joint task force. Cancer. 2014;120:2946-54.
- [Google Scholar]
- Anxiety, depression, and pain: Differences by primary cancer. Support Care Cancer. 2010;18:801-10.
- [Google Scholar]
- The validity of the hospital anxiety and depression scale. An updated literature review. J Psychosom Res. 2002;52:69-77.
- [Google Scholar]
- Screening for distress in cancer patients: A multicenter, nationwide study in Italy. Cancer. 2013;119:1714-21.
- [Google Scholar]
- Adjustment disorders: Fault line in the psychiatric glossary. Br J Psychiatry. 2001;179:479-81.
- [Google Scholar]
- The predictive value of adjustment disorders: a follow-up study. Am J Psychiatr. 1982;139:584-90.
- [Google Scholar]
- Differentiating major depression from adjustment disorder with depressed mood in the medical setting. Gen Hospital Psychiatr. 1990;12:159-165.
- [Google Scholar]
- Clinical relevance of adjustment disorder in DSM-III-R and DSM-IV. Comp Psychiatr. 1995;36:454-460.
- [Google Scholar]
- Adjustment reactions: A long term prospective and retrospective follow up of former patients in a crisis intervention ward. Acta Psychiatr Scand. 1991;84:86-93.
- [Google Scholar]
- International experiences with the hospital anxiety and depression scale – A review of validation data and clinical results. J Psychosom Res. 1997;42:17-41.
- [Google Scholar]
- Hospital anxiety and depression scale (HADS): Validation in a Greek general hospital sample. Ann Gen Psychiatry. 2008;7:4.
- [Google Scholar]
- Does the distress thermometer work? A systematic review of the evidence for its use and validity. BMJ Support Palliat Care. 2012;2:A30.
- [Google Scholar]
- The correlation of anxiety and depression levels with response to neoadjuvant chemotherapy in patients with breast cancer. JRSM Short Rep. 2011;2:15.
- [Google Scholar]
- Problems in cross-cultural use of the hospital anxiety and depression scale: “No butterflies in the desert”. PLoS One. 2013;8:e70975.
- [Google Scholar]
- Psychological factors associated with head and neck cancer treatment and survivorship: Evidence and opportunities for behavioral medicine. J Consult Clin Psychol. 2013;81:299-317.
- [Google Scholar]
- History of major depressive disorder prospectively predicts worse quality of life in women with breast cancer. Ann Behav Med. 2012;43:402-8.
- [Google Scholar]






