Translate this page into:
The Prevalence and Characteristics of Pain in Critically Ill Cancer Patients: A Prospective Nonrandomized Observational Study
Address for correspondence: Dr. Mayank Gupta; E-mail: drm_gupta@yahoo.co.in
This is an open access article distributed under the terms of the Creative Commons Attribution-NonCommercial-ShareAlike 3.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms
This article was originally published by Medknow Publications & Media Pvt Ltd and was migrated to Scientific Scholar after the change of Publisher.
Abstract
Context:
Pain is a distressing symptom common to all stages and ubiquitous at all levels of care in cancer patients. However, there is a lack of scientific literature on prevalence, severity, predictors, and the quality of pain in cancer patients admitted to an Intensive Care Unit (ICU).
Objectives:
To elucidate the prevalence of pain, moderate to severe pain, neuropathic pain, chronic pain, and pain as the most distressing symptom in critically ill-cancer patients at the time of ICU admission.
Methods:
We prospectively interviewed 126 patients within first 24 h of admission to a medical ICU. The patients were assessed for the presence of pain, its severity, sites, duration, nature, and its impact as a distressing symptom. Numerical Rating Scale and self-report version of Leeds Assessment of Neuropathic Signs and Symptoms were used to elucidate intensity of pain and neuropathic pain, respectively. Demographic characteristics such as age and sex, primary site, and stage of cancer were considered for a possible correlation with the prevalence of pain.
Results:
Of 126 patients included in the study 95 (75.40%), 79 (62.70%), 34 (26.98%), and 17 (13.49%) patients had pain, moderate-severe, chronic, and neuropathic pain, respectively. The average duration of pain was 171.16 ± 716.50 days. Totally, 58 (46.03%) and 42 (42.01%) patients had at least one and more than equal to 2 neuropathic pain symptoms, respectively. The primary malignancies associated with the highest prevalence of pain were genitourinary, hematological, and head and neck whereas breast and lung cancers were associated with the highest prevalence of neuropathic and chronic pain, respectively.
Conclusion:
The prevalence of pain among critically ill-cancer patients is high. Assessment for pain at the time of ICU admission would ensure appropriate assessment for the presence, type, severity, and the significance imparted to it.
Keywords
Cancer
Critically ill
Intensive Care Unit
Neuropathic
Pain
Severe pain
INTRODUCTION
Pain is ubiquitous and the most feared symptom amongst cancer patients.[1] An estimated 53% and 64–90% of patients with cancer and advanced cancer, respectively, suffer from pain; with more than one-third of patients having moderate-severe pain.[23] Extrapolating from these the prevalence of pain among critically ill cancer patients is expected to be high; however, there is a lack of convincing data particularly in Indian population. Moreover, there is a dearth of literature on characteristics and predictors of pain in this subgroup of cancer population. Systematic assessment of pain in cancer patients irrespective of the stage is mandatory in order to make correct estimates of pain prevalence and type.[4] Impeccable pain assessment and management is also imperative as it has been found to have a survival advantage in cancer patients. Tumor burden, concurrent infections, sepsis, the cumulative toxic effect of anti-cancer treatments and co-morbidities play havoc upon cancer patient's physiological homeostasis often requiring Intensive Care Unit (ICU) admissions. The multisystem involvement and gross physiological perturbations in cancer patients presenting to the ICU often shifts the management focus toward advanced life support measures, providing fertile ground for opiophobia to nurture, and symptomatic relief to take a backseat.[5] The patients themselves may not always volunteer pain information in an ICU setting Misconceptions based upon family and society perceptions that pain is an integral and indispensable component of the disease process also contribute toward silent suffering of the patients; a concept more imperative in a country like India where society plays a dominant role in influencing concepts regarding health and disease. The time of ICU admission usually provides the “golden hour” to assess the patient for subjective symptoms such as pain simultaneously/before initiation of the rigorous intensive management. This ensures the medical staff to become cognizant of presence and type of pain in accordance with patients own pain threshold, significance imparted to it and therapeutic end points before on-going physiological deviations makes the patient incoherent and incomprehensible. The present study aimed to determine the prevalence of moderate-severe pain, neuropathic pain, chronic pain, and “pain as the most distressing symptom” in cancer patients admitted to a medical ICU of a tertiary referral cancer hospital of Northern India.
MATERIALS AND METHODS
After obtaining ethical approval from the Institutional Review Board and written informed consent from the participants, all cancer patients admitted to the medical ICU of our hospital over a 3 months period were screened and 126 eligible patients were included in the study.
Inclusion and exclusion criteria
The inclusion criteria included:
-
Adult patient ≥18 years of age,
-
Patients diagnosed with cancer
-
Patients aware of their diagnosis and
-
Patients/relatives giving written informed consent for inclusion in the study.
Patients on mechanical ventilation with no cognitive impairement and able to respond in sign language such as “head nodding” or “pointing” were also included in the study. Patients with cognitive impairment, those not able to respond either verbally or in sign language were excluded from the study.
Data collection
A structured interview consisting of information regarding the presence of pain (Do you have pain right now?), its intensity, site, duration, and type was recorded by the duty doctor either at the admission or within first 24 h after initial clinical stabilization of patient's condition. The significance imparted to the pain was assessed by asking open-ended questions such as “Enumerate the most distressing symptom bothering you in the past 1-week?” and “does pain interfered with your sleep in the past 1-week?” A patient record form (PRF) to collect other pertinent data such as, age, sex, primary cancer site, stage, and time since diagnosis was also developed for the study. The structured interview and PRF were filled by different health care professionals unaware of each other's findings.
Tools and definitions
“Numerical Rating Scale” (NRS: 0 - no pain, 10 - worst possible pain) was used to rate “how much the pain hurts right now,” “worst,” and “average pain in the last 1-week” and were classified into:
None: 0
Mild: 1–3
Moderate: 4–6 and
Severe pain: ≥7.[6]
The patients were asked to mark/indicate all the sites of pain on a body diagram. The self-report Leeds Assessment of Neuropathic Signs and Symptoms (S-LANSS) pain scale in an interview format was administered to identify the presence of neuropathic pain’ (S-LANSS score ≥12).[7] The patients were also enquired for presence of various neuropathic pain descriptors such as “burning,” “tingling,” “electric shock-like,” “pressing,” and “pain evoked by light touching or pressure (otherwise nonpainful).” The patients were made to either mark or indicate their sites of pain on a body map. Patients with loco-regional progression, unresectable cancer, distant metastasis, on palliative therapy and those in whom anti-cancer treatment was no longer feasible were considered to have an advanced malignancy. Patients with potentially reversible physiological derangement involving two or more organ systems were considered to have multi-system organ dysfunction (MOD).
Outcomes
Our primary outcomes were the presence of pain (yes/no), moderate-severe (yes/no), chronic (yes/no), and neuropathic pain (yes/no). Our secondary outcomes were the presence of at least one neuropathic symptom, pain as the most distressing symptom and more than equal to 3 sites of pain.
Statistical analysis
The descriptive statistics of all these data were calculated in terms of frequencies and percentage of categorical variables. Mean (range: minimum-maximum) or average ± standard deviation has been used for the continuous variable (age, S-LANSS score).
RESULTS
A total of 126 consecutive eligible patients were included in the study [Figure 1]. The majority of the participants were females (56.35%) and in the 48–67 years age group (58.73%). The median age was 55 years (range: 18–82 years). The distribution according to the age and primary cancer site is shown in Tables 1 and 2, respectively. A total of 89 (70.63%) and 37 (29.37%) patients had advanced and early malignancy, respectively, and 68 (53.97%) patients had MOD [Table 3]. The most common reason for admission to the ICU included acute respiratory failure, shock, sepsis, altered sensorium, bleeding, and hypoglycemia.
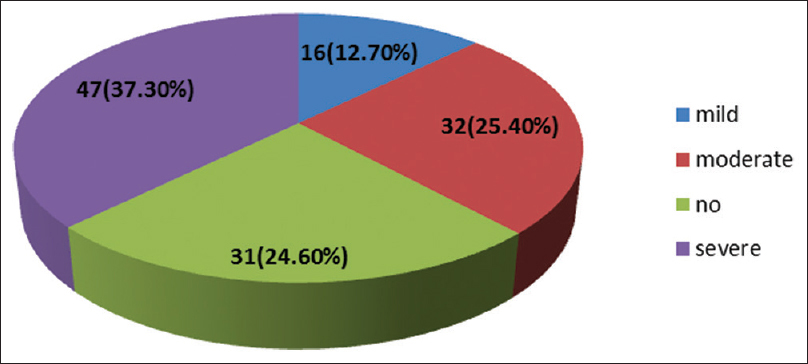
- Severity of pain



In the total study, population 75.4% of the patients had pain. The prevalence of pain, moderate-severe (NRS ≥ 4), chronic (duration ≥3 months), and neuropathic pain (primary outcomes) according to the primary cancer site is listed in Table 4. Demographic characteristics such as age and sex, primary cancer site, and cancer stage were considered for a possible association with prevalence of pain [Tables 3 and 5]. About 69.4% of the total study population of 126 patients had an advanced malignancy.


The mean duration of pain was 171.16 ± 716.50 days. The prevalence of pain according to different time durations (<1-week, 1-week to 1-month, >1-month to <3 months, ≥3 months to < 1-year, ≥1-year to 3 years, and ≥3 years) is depicted in Figure 2. A total of 49.2% patients reported pain to be the most distressing symptom at the time of ICU admission and 80% (76) of the patients having pain reported to have “pain interfering with their sleep.” The median S-LANSS pain score in patients having neuropathic pain was 17 (range: 13–24). The prevalence of patients with various neuropathic symptoms and number of neuropathic pain symptoms are shown in Table 6 and Figure 3, respectively. About 13.94% (17) patients had pain in more than equal to three body parts [Figure 4].

- Duration of pain

- Number of neuropathic pain symptoms

- Number of sites of pain

DISCUSSION
Inadequate pain assessment is one of the most common causes for inadequate cancer pain management and in turn contributes to poor quality of life.[89] The magnitude of unrelieved pain can be judged from the fact that pain has been declared as the “fifth vital sign.” An evaluation process based upon patient's own description of pain, its characteristics and significance imparted to it is an indispensable component of adequate cancer pain management. A high prevalence of pain (75.4%) was found in our study with 62.7% of the study population having moderate-severe pain. Our results are in accordance with other authors who have found the similar prevalence of pain and moderate-severe cancer pain ranging from 52–95% to 20–65%, respectively.[9101112131415] Cancer pain might perpetuates or act as a progenitor for other symptoms such as sleep disturbance, fatigue, and mood disturbances exacerbating the overall symptom burden.[1617] A total of 61.1% of the study population and 80% of the subset having pain reported it to interfere with their normal sleep cycle in our study. Earlier studies have also reported a similar magnitude of sleep problems (up to 72%) in cancer patients.[18] Such a combination of symptoms (pain, fatigue, and sleep disturbance) which tends to co-occur have been labeled as “symptom cluster” and it has been postulated that an intervention targeting one might benefit others as well.[1920] It is therefore imperative to shift the focus of management strategy from a single symptom to the symptom cluster as a whole. Acknowledging the coexistence and interrelation of these symptoms as in our study is the first step of this management strategy. In the current study, 49.2% of the patients reported pain to be the most distressing symptom. An awareness of the contribution of a pain to the overall patient's suffering will enable a physician to prevent the potentially preventable suffering due to pain from happening.[21] There is limited data available in the existing literature regarding the predictors of pain in cancer patients. The correlation between pain prevalence and cancer site is inconclusive at best. Some authors have not found any significant correlation while others have reported lung, head and neck, gastrointestinal, hematological, and others to have a higher prevalence of pain.[2222324] The malignancies associated with the highest pain prevalence in our study were genitourinary (85%), lung (81.25%), and head and neck (78.57%). However, only an overview and not definitive conclusions can be drawn regarding a positive correlation between pain prevalence and the tumor type because of the small number of patients with individual malignancies enrolled in the study. Studies to ascertain pain prevalence in individual tumor types are a way forward. The prevalence of pain was slightly higher in females compared to males (77.46% vs. 72.73%).
A number of tools and questionnaires (Neuropathic Pain Questionnaire, pain DETECT, Douleur Neuropathique 4 questions, ID pain) are available to assess neuropathic pain.[25] The S-LANSS was chosen because of its reliability and validity.[252627] Its sensitivity and specificity to diagnose NP has been found to be up to >90% compared to the clinical diagnosis.[252627] Neuropathic cancer pain (NCP) is often multifactorial in etiology with direct tumor infiltration, compression of nerves/plexus; chemotherapy/radiotherapy induced neuropathic pain, surgery-induced scarring or nerve damage as some of the most common causes.[28] Neuropathic pain was found in 13.49% and 46.03% of the patients reported the presence of at least one neuropathic pain symptom. Tingling followed by burning sensation were the most common neuropathic symptoms reported by the patients. In our study, 33.33% patients had more than one neuropathic symptom. Our results are in concordance with the available literature suggesting that a neuropathic pain component is present in 30% of cancer pain.[2829] No single sign or symptom is diagnostic of NCP. It is a combination of signs and symptoms that is more reliable. Paying attention to the pain descriptors used by the patient or probing for them helps in delineating the probable pathophysiology of pain, and hence, its appropriate management. The prevalence of NCP was the highest in breast cancer patients (41.67%). Breast cancer is frequently complicated by metastasis and bone involvement. Metastatic invasion of bony or nervous structures may be very painful and the activated osteoclast responsible for bone lysis also damage peripheral nerves leading to de-afferrentiation and neuropathic pain.[28] The neuropathic pain can also develop as a complication of breast surgery, the postmastectomy pain syndrome is known to occur in 20–68% of patients.[30]
Cancer patients frequently experience more than one type and sites of pain.[28] We found that 42.85% and 13.94% of the studied population had more than one site and more than equal to three sites of pain, respectively. The pathophysiology and etiology of pain at different sites in the same patient might be same or different. Therefore, it is imperative to ascertain the pain characteristics at all the sites of pain during initial and subsequent patient evaluation. In the current study, the average duration of pain was 171.16 days (range: 1–5110) and 26.98% of the patients had chronic pain ( ≥3 months). The prospects of “difficult to treat pain” and “NCP” increases with the chronicity of pain due to the possible neuroplasticity.[31]
Our study has important clinical implications. The results elucidate that there is a high prevalence of pain in critically ill-cancer patients at the time of ICU admission with the majority of them having moderate-severe pain. Our results also highlight that a large proportion of these patients have one or the other neuropathic pain symptoms. It is vital neither to take patient's silence as a surrogate for analgesia nor to consider pain in a complaining patient as an inseparable part and parcel of the disease state. Greater commitment and proactive measurement are required to alter the overall experience of seriously ill-patients.[32] It is the need of the hour to consider these palliative care needs simultaneously with regular ICU care.
Limitations
Our study had certain limitations. This study was an observational study. We did not ponder upon the pain management strategies and their benefit in terms of patient's satisfaction, morbidity, and mortality. The recognition is more critical as it lays the foundation stone for further management and research. Our study excluded the cognitively impaired patients. This was done intentionally to maintain uniformity in the study design and pain assessment tools used. This is not to imply the nonexistence of pain in these patients. Ascertaining the characteristics of pain in these patients is the next step ahead.
CONCLUSION
This study highlights that critically ill-cancer patients have a high prevalence of pain with a large proportion of them having moderate-severe and neuropathic pain. The time of ICU admission is the golden hour for assessment of the presence, type, severity, and the significance imparted to pain before on-going physiological perturbations makes them incoherent or incomprehensible.
Source of Support: Nil.
Conflict of Interest: None declared.
REFERENCES
- Stories of cancer pain: A historical perspective. J Pain Symptom Manage. 2005;29:22-31.
- [Google Scholar]
- Prevalence of pain in patients with cancer: A systematic review of the past 40 years. Ann Oncol. 2007;18:1437-49.
- [Google Scholar]
- Pharmacologic management of cancer pain. J Am Osteopath Assoc. 2005;105(11 Suppl 5):S21-8.
- [Google Scholar]
- NCCN practice guidelines for cancer pain. Oncology (Williston Park). 2000;14:135-50.
- [Google Scholar]
- Accuracy of the pain numeric rating scale as a screening test in primary care. J Gen Intern Med. 2007;22:1453-8.
- [Google Scholar]
- The S. LANSS score for identifying pain of predominantly neuropathic origin: Validation for use in clinical and postal research. J Pain. 2005;6:149-58.
- [Google Scholar]
- Prevalence and management of pain in Italian patients with advanced non-small-cell lung cancer. Br J Cancer. 2004;90:2288-96.
- [Google Scholar]
- Symptom distress in patients attending an outpatient palliative radiotherapy clinic. J Pain Symptom Manage. 2005;30:123-31.
- [Google Scholar]
- Nursing the late cancer patient at home; the family's impressions. Practitioner. 1959;183:64-9.
- [Google Scholar]
- Pain characteristics and treatment outcome for advanced cancer patients during the first week of specialized palliative care. J Pain Symptom Manage. 2004;27:104-13.
- [Google Scholar]
- Pain and its treatment in outpatients with metastatic cancer. N Engl J Med. 1994;330:592-6.
- [Google Scholar]
- High prevalence of pain in patients with cancer in a large population-based study in The Netherlands. Pain. 2007;132:312-20.
- [Google Scholar]
- Sensory and affective dimensions of advanced cancer pain. Psychooncology. 2002;11:23-34.
- [Google Scholar]
- Pain, sleep disturbance, and fatigue in patients with cancer: Using a mediation model to test a symptom cluster. Oncol Nurs Forum. 2005;32:542.
- [Google Scholar]
- Comparison of pain quality descriptors in cancer patients with nociceptive and neuropathic pain. In Vivo. 2007;21:93-7.
- [Google Scholar]
- Sleep-wake disturbances in people with cancer part I: An overview of sleep, sleep regulation, and effects of disease and treatment. Oncol Nurs Forum. 2004;31:735-46.
- [Google Scholar]
- Symptom clusters: Concept analysis and clinical implications for cancer nursing. Cancer Nurs. 2005;28:270-82.
- [Google Scholar]
- Mind-body treatments for the pain-fatigue-sleep disturbance symptom cluster in persons with cancer. J Pain Symptom Manage. 2010;39:126-38.
- [Google Scholar]
- Multidisciplinary management of cancer pain: A longitudinal retrospective study on a cohort of end-stage cancer patients. J Pain Symptom Manage. 2006;32:444-52.
- [Google Scholar]
- Prevalence of pain in patients with cancer: A systematic review of the past 40 years. Ann Oncol. 2007;18:1437-49.
- [Google Scholar]
- Status of cancer pain in Hanoi, Vietnam: A hospital-wide survey in a tertiary cancer treatment center. J Pain Symptom Manage. 2006;31:431-9.
- [Google Scholar]
- Identifying neuropathic pain in patients with head and neck cancer: Use of the Leeds Assessment of Neuropathic Symptoms and Signs Scale. J R Soc Med. 2003;96:379-83.
- [Google Scholar]
- Results of the Leeds assessment of neuropathic symptoms and signs pain scale in Turkey: A validation study. J Pain. 2004;5:427-32.
- [Google Scholar]
- Pharmacological treatment of neuropathic cancer pain: A comprehensive review of the current literature. Pain Pract. 2012;12:219-51.
- [Google Scholar]
- Refractory neuropathic pain: The nature and extent of the problem. Pain Pract. 2006;6:3-9.
- [Google Scholar]
- Neuropathic pain following breast cancer surgery: Proposed classification and research update. Pain. 2003;104:1-13.
- [Google Scholar]
- A controlled trial to improve care for seriously ill hospitalized patients. The study to understand prognoses and preferences for outcomes and risks of treatments (SUPPORT). The SUPPORT Principal Investigators. JAMA. 1995;274:1591-8.
- [Google Scholar]






