Translate this page into:
Burden and Outcomes of Pressure Ulcers in Cancer Patients Receiving the Kerala Model of Home Based Palliative Care in India: Results From a Prospective Observational Study
Address for correspondence: Dr. Santam Chakraborty; E-mail: drsantam@gmail.com
This is an open-access article distributed under the terms of the Creative Commons Attribution-Noncommercial-Share Alike 3.0 Unported, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
This article was originally published by Medknow Publications & Media Pvt Ltd and was migrated to Scientific Scholar after the change of Publisher.
Abstract
Aim:
To report the prevalence and outcomes of pressure ulcers (PU) seen in a cohort of cancer patients requiring home-based palliative care.
Materials and Methods:
All patients referred for home care were eligible for this prospective observational study, provided they were living within a distance of 35 km from the institute and gave informed consent. During each visit, caregivers were trained and educated for providing nursing care for the patient. Dressing material for PU care was provided to all patients free of cost and care methods were demonstrated. Factors influencing the occurrence and healing of PUs were analyzed using logistic regression. Duration for healing of PU was calculated using the Kaplan Meier method. P < 0.05 are taken as significant.
Results:
Twenty-one of 108 (19.4%) enrolled patients had PU at the start of homecare services. None of the patients developed new PU during the course of home care. Complete healing of PU was seen in 9 (42.9%) patients. The median duration for healing of PU was found to be 56 days. Median expenditure incurred in patients with PU was Rs. 2323.40 with a median daily expenditure of Rs. 77.56.
Conclusions:
The present model of homecare service delivery was found to be effective in the prevention and management of PUs. The high prevalence of PU in this cohort indicates a need for greater awareness for this complication.
Clinical Trial Registry Number:
CTRI/2014/03/004477
Keywords
Bed sore
Cancer
Decubitus ulcer
Home care
Palliative care
Pressure ulcer
HIGHLIGHTS
-
A prospective observational cohort study of patients presenting for home care
-
Almost 1/5th of patients with advanced cancers suffer from pressure ulcers
-
None of the patients enrolled had a new pressure ulcer during home care
-
Pressure ulcer resolved completely with care in 40% of the patients
-
This care could be provided at a median cost of Rs. 77 per day per patient (~ $ 1.2).
INTRODUCTION
The National Pressure Ulcer Advisory Panel (NPUAP) defines pressure ulcers (PU) as a form of “localized tissue injury that develop as a result of pressure or pressure in combination with friction/shear.”[1] The magnitude of this condition is variable. Hendrichova et al., from Italy had found a rate of 22.9% in cancer patients treated at a palliative care unit.[2] Brink et al., found a prevalence rate of 10.5% in patients under home care for terminal cancers.[3]
Management of PUs can be very expensive. Dealey et al., estimated that the cost of treating PUs in UK ranged between £ 1,214–14,108 depending on the severity of the ulcer.[4] The healthcare system in India is overburdened and few facilities are available for providing end-of-life care. Invariably, most of the care is delivered at home and caregivers are family members. The Kerala model of palliative care depends on a network of trained volunteers and nurses who provide homecare services through regular visits.[5] In addition to developing a trained cadre of community-based care providers, several innovative methods are used to bring down the cost of care.
Our homecare service consists of trained palliative care nurses who conduct regular homecare visits for terminal cancer patients supported by a trained palliative care physician. For logistic reasons, this is restricted to a distance of 35 km around the hospital. While there are few publications on the burden of PUs in Indian patients in hospital settings, we could not find any data in patients suffering from cancer.[67] In the present paper, we present data on prevalence of PU, the effectiveness in terms of healing, duration of persistence as well as the expenditure incurred for this care in a cohort of homecare patients with cancer included for a prospective study.
MATERIALS AND METHODS
After obtaining IRB approval, patients presenting for home care were included in this prospective observational study (CTRI Number: CTRI/2014/03/004477). Written informed consent was obtained from all participants prior to the first visit. All patients referred for homecare services were eligible for inclusion in the study. Patients were selected for homecare service provided they resided within a distance of 35 km from the center. Patients were recruited for the study between October 2010 till December 2013. Patients were followed up at fortnightly interval till death.
As a part of the homecare process, a trained nurse would visit the house of the patient and provide care and education. Visits were conducted as per a preplanned route plan at fortnightly intervals. During each visit, subjective and objective problems being faced were documented in a pre-specified proforma. The data collected included the size, location, and stage of PU if any. All PUs were staged by the nurse using the NPUAP staging scheme.[8] In patients without PUs, caregivers were trained in the importance of regular postural changes, maintaining hygiene and nutrition. During each visit, the trained nurse assessed if any PUs had developed in the interim. In patients with pre-existing PUs, similar education and training was provided. In addition, simple saline dressings were instituted using cloth strips cut from cotton dhotis. These strips were boiled in a steamer that is meant for steaming pancakes (idlis) for at least 20 minutes. Instead of using commercially available normal saline, normal drinking water mixed with 2 tsp salt per liter of water would be boiled for 5 minutes and used for cleansing. For patients with foul smelling/malodorous wounds metronidazole tablets would be crushed and applied on the wound to control anerobic growth.
The cost of medications and other consumables supplied to the patient as well as the cost of staff and salary was borne by the hospital. The fixed cost per visit which included payment for the nurses salary, driver salary, and car fuel costs worked out to be Rs. 455 per patient per visit. For the purpose of this calculation, the salary paid per hour was derived from the monthly salary for the driver and nurse. Each visit was assumed to take 1 hour time per patient. The total costs of consumable items like medications given to each patient were derived from the medicine records maintained prospectively for each visit. The cost of metronidazole tablets and sticking tapes used in patients with PU was included in the wound care costs. The total costs of visits were calculated by adding the total expense per visit with the expenses incurred in consumable items like medicines. The total cost was divided by the total number of days in home care to obtain the cost per day for each patient.
Data on demographic variables, cancer site, stage, treatment received, and performance status at first visit was recorded on a datasheet. Statistical analysis was done using the R software version 3.1.0. Median and interquartile range were calculated for continuous variables, while frequencies were calculated for categorical variables. Logistic regression analysis was conducted to find out the influence of various prognostic factors on the healing of PUs. Duration of persistence of PUs was estimated from the date it was noticed first till the date it was healed. In patients with PU at the first visit, the date of first visit was taken as the start date. Patients were censored at the date of death if the ulcer was not completely healed by then. New PUs developing in the same patient was recorded as a separate event. Factors influencing the duration of PUs were evaluated using the log rank test. P < 0.05 were taken as significant. For purpose of analysis, cancers with less than 10 patients were grouped together as a single category.
RESULTS
A total of 108 patients were included in this study. Table 1 shows the demographic profile of the patient population in the study. Out of these, 27 (25%) patients were unfit for any chemotherapy, surgery, or radiotherapy and were referred for palliative care. In these patients, 23 had progressive disease after receiving treatment for their cancer outside the hospital while 4 were not treated in view of their advanced age and performance status.
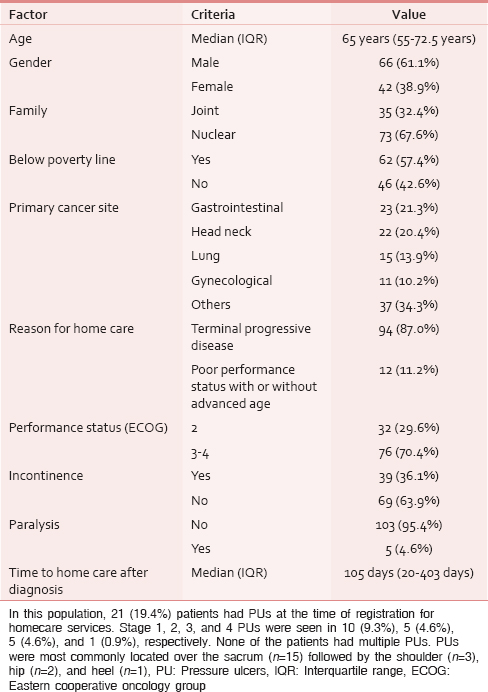
Nineteen (17.6%) patients had received curative intent treatment with surgery or radiotherapy while 62 (57.4%) had received palliative treatment prior to registration for home care. In the 62 patients treated with palliative intent after diagnosis, palliative chemotherapy had been delivered in 25 patients while the rest (n = 37) received palliative radiotherapy.
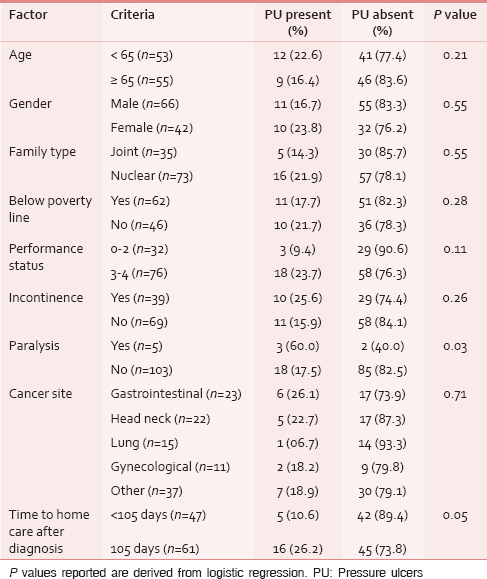
Table 2 shows the number of PUs stratified according to the risk factors present.
In 21 patients with PUs, complete healing of the PUs was seen in 9 (42.9%) while in another 5 (23.8%), reduction in the stage was observed. In 5 (23.8%) patients, the stage of PU did not change while in 2 (9.5%) patients, an increase in stage was noted. None of the 108 patients developed new PUs during the period of homecare visits. Figure 1 is a waterfall plot showing the change in the stage of PUs between the first and the last homecare visit. Both the 2 patients who had an increase in the stage of PU during homecare visits had an initial stage 1 PU that subsequently increased to stage 2 before they died.
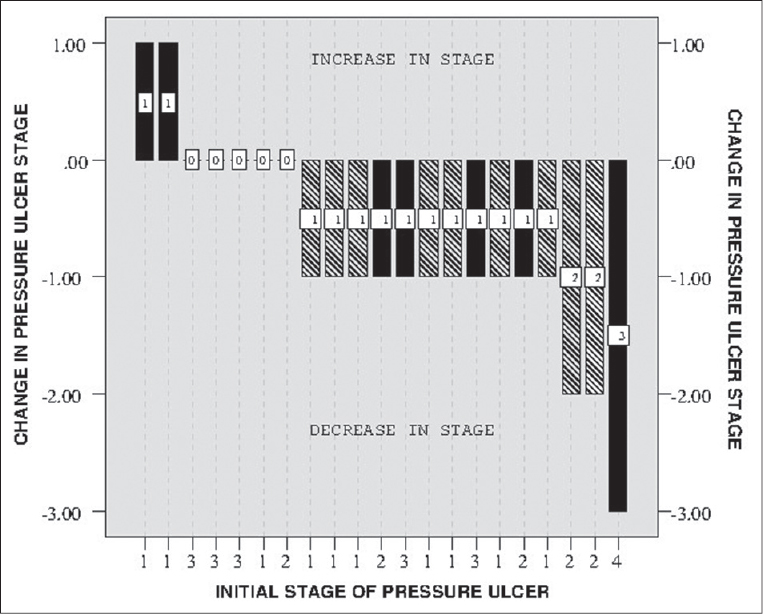
- Waterfall plot showing the extent of change in the stage of PU between the 1st and last homecare visit. Figures in X axis represent the stage of PU at the first visit. Patients with complete resolution of PU are depicted by hatched bars while solid bars represent patients where PUs did not resolve completely. As can be seen none of the patients with Grade 3–4 pressure ulcers had a complete resolution. However, in these patients, a decrease in stage was observed in 3 patients while in other 3 the stage remained stable
Table 3 shows the influence of various factors on the healing of PUs. On logistic regression analysis financial status, paralysis, and performance status significantly influenced the healing PUs.
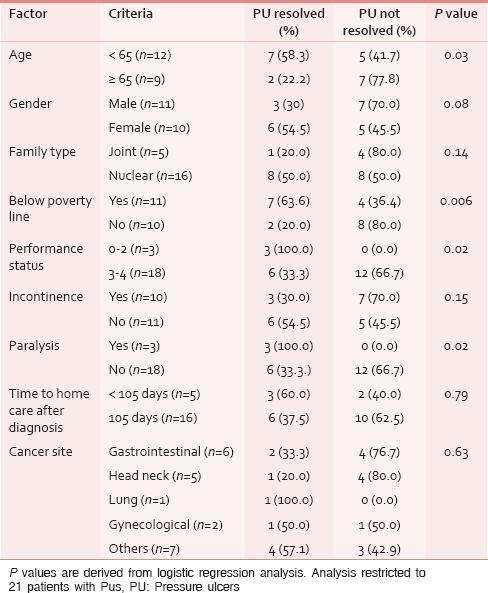
Median estimated duration of persistence of any PU in these 21 patients was 56 days (95% CI: 0–117 days) [Figure 2]. The median duration of survival of patients with stage 1–2 PU was 75 days versus 37.5 days for stage 3–4 PU (P = 0.22, NS). In the 21 patients with PU, any PU was observed by the homecare team in a median of 3.5 visits (IQR: 2.5–4.5). In this same group, the median total number of visits conducted was 4 (IQR 3.5–4.5).
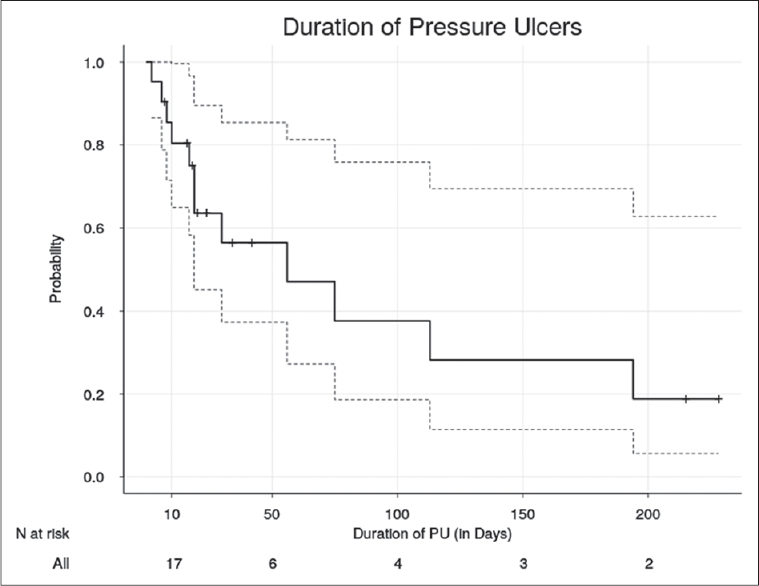
- Kaplan Meier curve showing the time of persistence of PU in 21 patients. Patients without PU healing are censored. Dotted lines show the 95% confidence intervals. Numbers at risk at each time point are shown below the graph
The median total expenditure incurred in caring for the patients with PUs was Rs. 2323.40 (IQR: Rs. 1878.40–2768.40) and the median daily expenditure was Rs. 77.56 (IQR: 99.86–55.26). This includes the cost of homecare visits where care for PU had to be provided as well as visits were PU care was not provided as the PU had healed. The median total cost incurred for wound care was Rs. 96.60 (IQR: Rs. 21.60–171.60). The median total cost of the medications was Rs. 470.00 (IQR: Rs. 174.18–765.82).
The total cost incurred by the hospital in caring for patients in the homecare services was Rs. 2,01,368.50. Out of this, Rs 60,254.00 was spent on medications. The expenditure incurred by the hospital in caring for the 21 patients with PU was Rs. 58,193.00 with the cost of medications coming to Rs. 17179.00. The total cost incurred on the wound care for these patients was Rs. 1883.70 only.
DISCUSSION
The present study showed that almost 20% patients with advanced cancer taken up for homecare services suffer from PU in our population. This figure is comparable to that reported by Hendrichova et al.,[2] but higher than that reported by Brink et al.[3] This high prevalence of PU at presentation points to the need for sensitization of oncologists towards this common but unappreciated problem faced by patients with terminal cancers.
Poor performance status and presence of paralysis predicted an increased risk of presenting with PUs in this cohort of patients [Table 2]. The causes behind development of PU is poorly understood and is not a result of a single factor. In a recent systematic review Coleman et al., have identified three major domains that predict an increased risk of PU development.[9] These domains are mobility/activity, skin perfusion (e. g., diabetes) and skin condition, particularly existence of a stage I PU, as important predictors of development of PU. Due to the small number of patients with PU, only the single domain of reduced mobility/activity (in form of paralysis and performance status) emerged as significant. The limited lifespan of these patients also influenced this as parameters related to limited skin perfusion and existing PU are likely to be significant only with longer duration of follow-up.
In the present study, 40% of the PUs had completely healed before the last homecare visit. The median time taken for complete healing of pressure ulcers was 56 days from the date of registration in homecare services. This is comparable to the results reported by McNees et al., where 44% of pressure ulcers and wounds had healed in cancer patients and the median time taken for healing was 55 days (±49 days).[10] In contrast, as reported by Hendrichova et al., in their study only 10% of pressure wounds had healed prior to the death of the patient.[2]
In contrast to stage I and II PUs, majority of which healed in the present study, none of the stage III-IV PU healed completely. However, 50% of these patients had a reduction in stage and none had a worsening in the stage. Brandeis et al., have shown that with good nursing care majority of the pressure ulcers heal within a period of 1 year.[11] In the present cohort, the limited lifespan of the patients with advanced pressure ulcers (median survival with stage 3–4 ulcer: 37.5 days only) prevented complete healing. As reported by Brown et al., in patients surviving less than 180 days (6 months), none of stage IV PUs healed completely.[12] In fact the authors hypothesized that such PUs may represent a reflection of the underlying disease burden and be a part of the spectrum of the overall co-morbidity preceding death in such patients.
The healing of the PUs in the present study was influenced by the baseline performance status and presence of paralysis. While patients with poorer performance status had a poorer chance of healing of PU, paradoxically in all three patients with paralysis the PS healed. Out of these three patients, two had hemiplegia and one had paraplegia. Two patients had a stage I PU and one had a stage II PU all three of which had healed completely by the end of the homecare visits. This finding is likely to be due to chance because of the small number of patients.
The model of homecare service as delivered through our center is a derived from the Kerala model of home-based palliative care services.[5] An advisory model is adopted and patient's relatives are entrusted with providing nursing care without relying on hiring specialized nurses. This model was borne out of necessity, as there is a triple combination of high patient load, lack of trained nursing manpower, and poor financial status in our part of the world. In developed nations, the cost of caring for PUs alone can exceed $ 2 billion (~1% of total healthcare budget) as reported by Severens et al.[13] Such expenditure is not possible in our country where the total healthcare budget is only $ 6 billion. Additionally, the nature of family ties in our part of the world is such that usually want to take care and nurse the sick patient. The role of the homecare team is to educate and empower the caregiver in the family by teaching them simple and cost effective methods of caring for the patients, as well as allaying their apprehensions and misgivings.
However in most such palliative homecare services, trained physicians are not available. As a result in the event of any untoward event relatives are forced to turn to local hospitals where patients may not have been primarily treated for their cancer. The present homecare team on the other hand has a close communication with the palliative care specialist as well as other oncologists in the hospital. As a result, in the event of any emergency or sudden problem, patient is assured of specialist support as required. Thus both the patients as well as the caregivers are assured of continuity of care.
The present study is thefirst study which reports the prevalence and outcomes of PUs in terminal cancer patients managed using the Kerala model of palliative home care. The strengths of this study are its prospective nature wherein detailed observation of the patients’ condition was made by a trained palliative care team. The low cost of the present model is evident in the fact that the total expenditure for caring of 21 patients with PU was Rs. 58,193.00 which works out to be approximately $1000.00. This compares very favorably to the cost incurred for caring and preventing PU in a long term care facility in 539 patients as reported by Xakellis et al.[14]
Due to resource limitations, we could only include patients who resided within a distance of 35 km from the institute. Given the hybrid nature of our homecare model, the outcome data cannot be readily generalized to patients referred to other homecare services in India. It is also unknown if the effectiveness of care as used in this model compares favorably to the more established and expensive methods of wound care. The costs reported are direct costs incurred by the hospital as indirect costs incurred by the patients relatives in caring for the patients with PU were not recorded. The actual indirect cost of such a care model needs to be further studied using prospective studies. In this study, no dedicated risk assessment for PU was done using a standard instrument. However, a recent Cochrane review has also shown that there is little evidence that use of structured PU risk assessment tool is useful in reducing the risk of developing PU.[15]
CONCLUSION
Our prospective study demonstrates that there is a high prevalence of PUs in patients referred to home care with terminal cancers. The hybrid model of homecare service delivery, as adopted in the present study, was able to prevent new PUs from developing in the patient population. In addition, complete healing was seen in 40% of the patients, with an average expenditure of Rs. 2323.40 (~ $40) per patient with PU.
Source of Support: Nil.
Conflict of Interest: None declared.
REFERENCES
- National Pressure Ulcer Advisory Panel. Pressure ulcers in individuals receiving palliative care: A National Pressure Ulcer Advisory Panel white paper. Adv Skin Wound Care. 2010;23:59-72.
- [Google Scholar]
- Factors associated with pressure ulcers in palliative home care. J Palliat Med. 2006;9:1369-75.
- [Google Scholar]
- The cost of pressure ulcers in the United Kingdom. J Wound Care. 2012;21:261-2. 264, 266
- [Google Scholar]
- Kerala, India: A regional community-based palliative care model. J Pain Symptom Manage. 2007;33:623-7.
- [Google Scholar]
- The prevalence of pressure ulcers in hospitalised patients in a university hospital in India. J Wound Care. 2005;14:36-7.
- [Google Scholar]
- Risk assessment for pressure ulcer: A hospital-based study. J Wound Ostomy Continence Nurs. 2008;35:407-11.
- [Google Scholar]
- National Pressure Ulcer Advisory Panel. National Pressure Ulcer Advisory Panel's updated pressure ulcer staging system. Adv Skin Wound Care. 2007;20:269-74.
- [Google Scholar]
- Patient risk factors for pressure ulcer development: Systematic review. Int J Nurs Stud. 2013;50:974-1003.
- [Google Scholar]
- Pressure ulcers and other chronic wounds in patients with and patients without cancer: A retrospective, comparative analysis of healing patterns. Ostomy Wound Manage. 2007;53:70-8.
- [Google Scholar]
- The epidemiology and natural history of pressure ulcers in elderly nursing home residents. JAMA. 1990;264:2905-9.
- [Google Scholar]
- Long-term outcomes of full-thickness pressure ulcers: Healing and mortality. Ostomy Wound Manage. 2003;49:42-50.
- [Google Scholar]
- The cost of illness of pressure ulcers in The Netherlands. Adv Skin Wound Care. 2002;15:72-7.
- [Google Scholar]
- The cost of healing pressure ulcers across multiple health care settings. Adv Wound Care. 1996;9:18-22.
- [Google Scholar]
- Risk assessment scales for pressure ulcer prevention: A systematic review. J Adv Nurs. 2006;54:94-110.
- [Google Scholar]






