Translate this page into:
Hypofractionated Palliative Radiotherapy in Locally Advanced Inoperable Head and Neck Cancer: CMC Vellore Experience
Address for correspondence: Dr. Saikat Das; E-mail: saikat@cmcvellore.ac.in
This is an open-access article distributed under the terms of the Creative Commons Attribution-Noncommercial-Share Alike 3.0 Unported, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
This article was originally published by Medknow Publications & Media Pvt Ltd and was migrated to Scientific Scholar after the change of Publisher.
Abstract
Background:
A novel, short duration, palliative radiotherapy schedule for inoperable head and neck cancer was evaluated in terms of palliation of cancer-related symptoms and acute toxicities.
Materials and Methods:
Thirty-six patients with inoperable head and neck cancer were included in the study (2010-2012). All patients received 40 Gy in 10 fractions (equivalent dose: 49.8 Gy in conventional fractionation) with 2 fractions per week. Treatment-related toxicity was assessed using Radiation Therapy Oncology Group criteria. Functional Assessment of Cancer Therapy (Head and Neck, FACT H and N) quality of life (QOL) tool was administered before starting and at the completion of radiotherapy. Mean value before and after treatment was compared (paired t-test, P = 0.05, two-tailed for significance).
Results:
Thirty-three patients (male: 29, female: 4, mean age: 57.8 ± 9.7 years) were included in the analysis (three patients discontinued treatment due to socioeconomic reasons). All patients had advanced inoperable head and neck cancers (27% IVA, 61% IVB, 9% IVC, TNM stage and 3% recurrent disease). Distressing pain at primary site (42%), dysphagia (18%), neck swelling (30%), and hoarseness (10%) were common presentations. Incidence of grade III mucositis and dermatitis and pain was 18%, 3%, and 24%, respectively. Planned radiotherapy without any interruptions was completed by 73% patients. QOL assessment showed improvement in social well-being (17.4 vs. 20.01, P = 0.03), but no significant change was observed in head and neck specific score (25.1 vs. 25.0, P = NS) after treatment. Reduction of pain was observed in 88% patients and 60% patients had improvement of performance status. Median overall survival of the cohort was 7 months.
Conclusions:
The study shows that this short duration palliative radiotherapy schedule is a clinically viable option for advanced inoperable head and neck cancer to achieve significant palliation of the main presenting symptoms like pain, dysphagia, and throat pain.
Keywords
Cancer pain
Hypofractionated radiotherapy
Inoperable head and neck cancer
Palliative radiotherapy
INTRODUCTION
Head and neck cancer is one of the highly prevalent cancers in developing countries like India.[12] Population-based cancer registry in India projects that the number of tobacco- related cancer and head and neck cancer would be 3, 16, 734 and 2, 18, 421, respectively, by 2020.[3] In most cases, due to extensive locoregional involvement, poor general condition of the patient, or comorbid conditions curative treatment is not possible. Therefore, the relevance of aggressive treatment in unresectable locally advanced head and neck cancer has been questioned.[4567] The intent of treatment in such cases is to improve the quality of life of the patients keeping their socioeconomic condition in mind and judiciously utilizing the precious resources for curable conditions.
The epidemiology and biology of head and neck cancer in developing countries are different from that of developed countries.[4] Therefore, the treatment data from Western literature should be extrapolated with caution in developing countries like India. The first report on the experience of treatment of buccal mucosa cancer in India was published from our institute in 1966 by Singh et al.[89] The study showed about 30% of the cases was in very advanced stages and a large percentage of patients (60%) did not have any treatment at all due to various socioeconomic reasons. During that period, treatment with radiotherapy and or chemotherapy resulted in a response rate of 19% at 3 years.
With this background, we report the feasibility of a novel, short duration, hypofractionated, palliative radiotherapy schedule for locally advanced inoperable head and neck cancer for palliation of cancer-related distressing symptoms. Occurrence of acute treatment-related toxicities and improvement in quality of life (based on FACT-HN scale) at completion of radiotherapy compared to baseline is also reported.
MATERIALS AND METHODS
Thirty-six patients with inoperable, head and neck cancer not fit for radical treatment were prospectively treated with a short course of radiotherapy (2010-2012) after obtaining informed consent.
Inclusion criteria
-
Surgical unresectable stage IVB head and neck cancer (extension to infratemporal fossa extension, carotid space invasion, prevertebral fascia invasion, fixed-fungating nodal mass)
-
Stage IVA disease-poor performance status, significant comorbid illness
-
Stage IVC disease-for palliation of local symptoms
-
No previous history of radiotherapy/chemotherapy
-
Nonnasopharyngeal, non-PNS, and nonsalivary gland primary
-
No previous history of cancer.
Radiation planning and treatment
All patients received 40 Gy in 10 fractions (equivalent dose: 49.8 Gy in conventional fractionation) with 2 fractions per week (Monday and Thursday or Tuesday and Friday, equivalent weekly dose 10 Gy). Patients were immobilized in supine position and head fixed with thermoplastic ray cast. Primary and nodal gross tumor volumes were outlined based on clinical examination, ENT examination including nasopharyngolaryngo endoscopy. Radiotherapy portal included gross tumor volume with 2 cm margin and the high-risk nodal regions. Prophylactic radiation of anterior neck was not done. Radiotherapy was delivered by parallel opposed lateral technique in telecobalt machine (Theratron 780C). Spine shielding was done at 32 Gy, if the nodal volume was situated anterior to the spine. If the nodal volume was posterior to the spine, shielding was omitted [Figure 1].
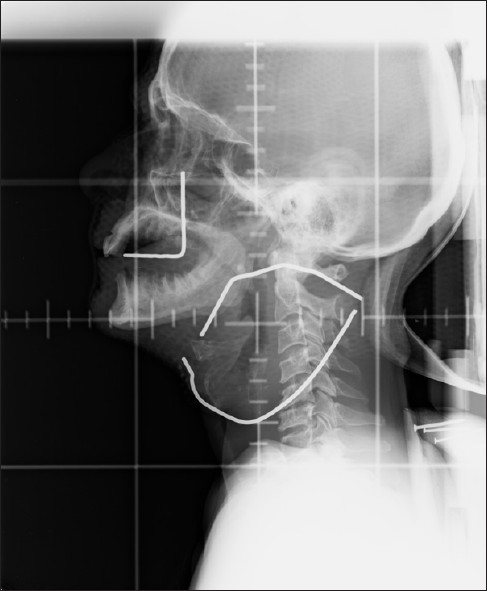
- Simulator film of a patient: 69-year-old gentleman, a chronic smoker and a known case of Hansen's disease evaluated for neck swelling and hoarseness of voice for 6 months. ENT examination revealed a slough covered growth in the right pyriform sinus extending to the arytenoids, aryepiglottic fold up to the left arytenoids. Fine needle aspiration cytology from the node was reported as metastaic squamous cell carcinoma and was staged T2N3M0 (stage IVB disease inoperable stage) was planned for palliative radiotherapy. The node has been outlined with lead wire during simulation
Supportive care during radiation
Distressing presenting symptoms like pain, painful ulcer, dysphagia, odynophagia, breathing difficulty and neck swelling was recorded and graded on a pragmatic scale of mild, moderate, and severe as per the patient's reporting of the complaint. All patients received nutritional advice by dietician and nasogastric feeding was offered if required.
Assessment of toxicity and quality of life
Patients were assessed weekly for assessment of treatment-related toxicity and tolerance. Treatment-related toxicity including radiation mucositis, dermatitis was assessed using RTOG criteria.[10] Patients were specifically asked about symptom relief compared to baseline presentation. Quality of life was assessed by Functional Assessment of Cancer Therapy (Head and Neck, FACT H and N) quality of life tool. This is a validated and accepted QOL tool for head and neck cancer. This was administered before starting and at the completion of radiotherapy.
Posttreatment follow-up
Tumor response and symptom relief was periodically monitored at 1 month and then 3 monthly or earlier if necessary. All patients were initiated with tyrosine kinase inhibitors (tab. gefitinib 250 mg once daily), 1 month after completion of radiotherapy till disease progression. Patients with progressive disease were treated with symptomatic care only.
Statistical analysis
Primary outcome (categorical variable) was compared by frequency distribution. Overall survival was assessed by Kaplan-Meier survival estimate. Toxicity was graded (categorical variable) and compared the frequency distribution between groups.
Mean value of physical, social, emotional, functional, and head and neck-specific quality of life before and after treatment was compared by paired t-test (P = 0.05, for significance).
RESULTS
Patient, tumor, and treatment-related characteristics
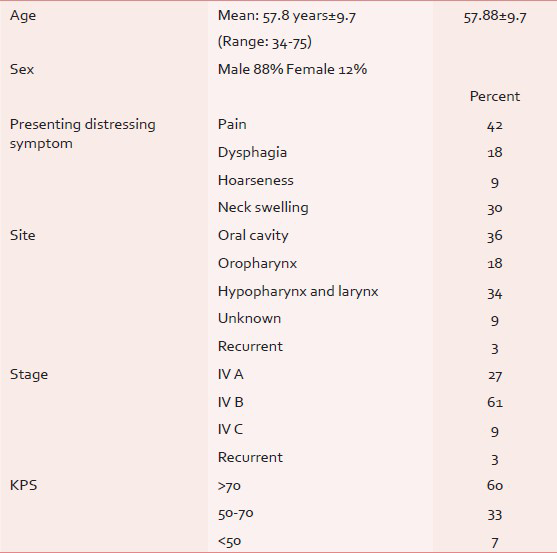
Radiotherapy records of total 33 patients were reviewed and included in the analysis (88% male and 12% female). Three patients discontinued treatment due to socioeconomic reasons. The mean age was 57.88 years (Range: 34-75 years). About 24% patients did not have any formal education and 52% possessed an educational qualification of high school and above. The median performance status as measured by Karnofsky performance scale was 70. About 72% of the subjects belonged to low socioeconomic status as per modified Kuppuswamy index of socioeconomic status. In the study group, 85% was addicted to some form of tobacco. Among them, 55% were addicted to smoking and 6% to chewable tobacco, 21% to both smoking and alcohol.
The commonest presenting symptom was a pain (42%) which includes pain in the throat, painful ulcerative lesions, or referred pain to ear. Dysphagia was present in 18% patients. About 30% patients presented with neck swellings. Oral cavity tumors were the commonest site (36%), followed by larynx, hypopharynx (34%), and oropharynx (18%). Most of the patients had stage IV tumor (97%, IV A, IV B, IV C in 27%, 61%, and 9%, respectively) [Table 1]. Histologically, 52% of the tumor was moderately differentiated, 15% well-differentiated and 12% poorly differentiated and 21% being metastatic carcinoma based on cytology from neck node.
Treatment-related toxicities and feasibility
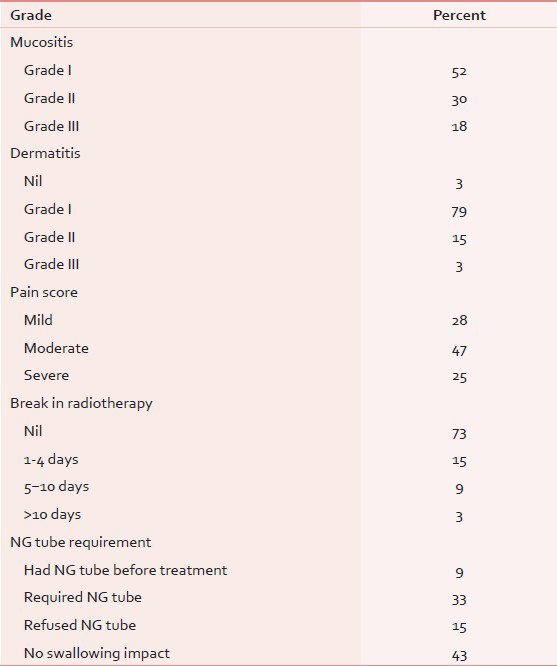
The radiotherapy schedule was well-tolerated [Table 2]. About 73% of the patients completed the planned radiation therapy without any break during the treatment. As per the assessment of toxicity (RTOG) grade 3 mucositis and dermatitis occurred in 18% and 3%, respectively. One-fourth of the patients experienced severe pain which was treated as per the WHO pain ladder. Only two patients (6%) needed morphine for pain relief during the course of radiotherapy. Remaining patients had only mild to moderate pain. About 43% patients did not have any swallowing impact before or during the treatment. Nasogastric tube was placed before radiotherapy in three patients (9%), and five patients refused to have nasogastric tube even after being advised. One patient, who had an extensive buccal mucosa tumor with orocutaneous fistula, had worsening of symptoms (pain, difficulty in swallowing) during radiation therapy leading to a break in the treatment for nearly 15 days. She was hospitalized and managed with best supportive care, following which she completed the treatment uneventfully. Three patients needed hospitalization during the course of treatment. Median duration of hospitalization was 6 days.
Improvement of key performance indicator
At the end of the radiation therapy, significant pain relief (more than 50%) was obtained in about 88% of patients and worsened in 9% subjects at the end of radiotherapy. Morphine requirement for pain relief after completion of treatment was 12%. The performance status improved in 60%, did not change in 33%, and deteriorated in about 7% of patients. Treatment resulted in improvement of swallowing in 46% patients and almost a similar number of patients had no improvement. Worsening was observed in nearly 5% of the population.
Quality of life analysis
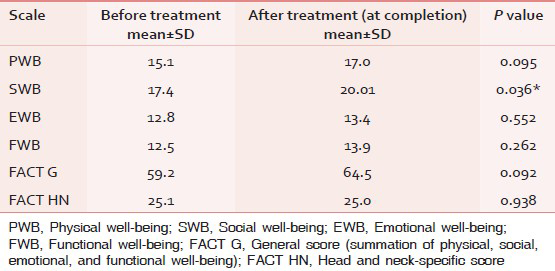
There was improvement in all the aspects of the quality of life (physical, social, emotional, and functional well-being) at the end of radiotherapy. This is shown in Table 3. A statistically significant improvement in social well-being was noted after treatment (17.4 vs. 20.01, P = 0.03). No significant change was observed in head and neck-specific score after treatment (25.01 vs. 20.0).
Survival analysis
Median follow-up period of the cohort was 6 months (1-26 months). The median survival was 7 months (4-9 months). At 2 years, cumulative survival probability was 17% [Figure 2].
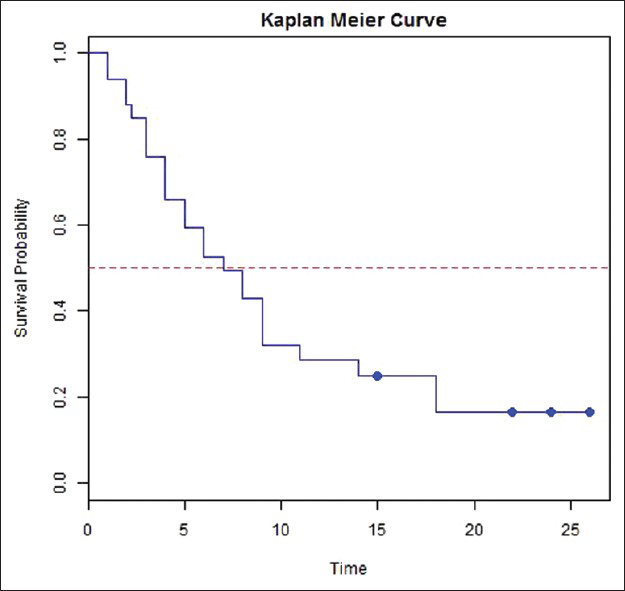
- Overall survival and Kaplan-Meier curve (median survival indicated by the dotted line was 7 months)
DISCUSSION
Locally advanced head and neck cancer constitutes about 25% of cancer burden in clinical practice in developing countries like India.[11] Response of inoperable locally advanced head and neck cancer to aggressive treatments including chemoradiotherapy is poor and is associated with significant treatment-related toxicities.[12] In India, about 70-75% cases of head and neck cancer present in a locally advanced stage with a significant portion in an inoperable stage.[13] Palliation of distressing presenting symptom like painful ulcer, throat pain, swallowing difficulty, and breathing difficulty is the main objective of treatment,[14] with minimal treatment-related toxicities. Instead of increasing the life expectancy, improvement of quality of life and cost benefit issues are more important in our settings.[12] In India, most of the tertiary cancer centers like that of ours are overloaded with patients.
In order to strike a balance between radiobiologically effective dose and overall treatment duration, the present dose fractionation scheme was selected. Total dose selected was based on the only randomized study of palliative radiotherapy in advanced head and neck cancer.[15] The twice weekly treatment was designed to reduce the number of hospital attendance as suggested by the “Hypo Trial.”[16] The scheme had a high patient compliance rate. It also had the advantage of less opportunity of tumor repopulation.
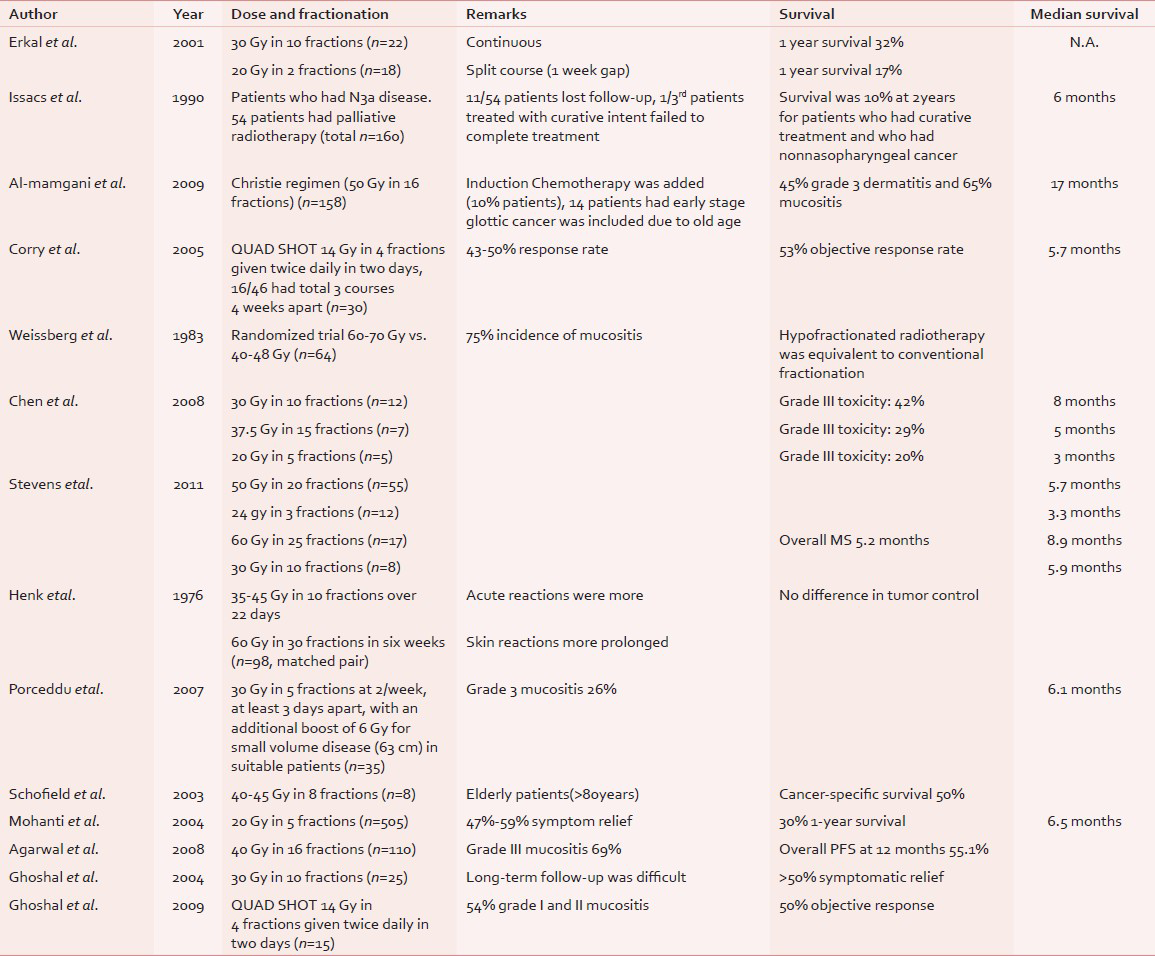
An optimal dose fractionation schedule for palliative radiotherapy in head and neck cancer is yet to evolve even though there are some guidelines for curative settings.[17] Current evidence seems to favor short course palliative radiotherapy schedule than single fraction or protracted course of radiation.[11] Historically, during the World War II, 3-5 weeks regimen was shown to be superior to 1-4-8 days regimen (28% vs. 3% control rate at 3 years for laryngopharyngeal cancer) in Christie Hospital, Manchester[18] as highlighted by Paterson in the Mackenzie Davidson Memorial lecture in 1952. Patients treated with palliative intent decision on dose and fractionation is often based on the feasibility, quality of life, and palliation rather than on survival or radiobiological considerations.[19] Various hypofractionated palliative radiotherapy schedules have been reported in the literature from Western countries as well as from India. Table 4 gives a detailed review of the palliative radiotherapy schedules for locally advanced head and neck cancer and their major findings.[7151619202122232425] One randomized study reported by Weissberg et al.,[15] compared conventionally fractionated radiation of 60-70 Gy (over 6-7 weeks) with 40-48 Gy (over 2-3 weeks). Hypofractionated schedule was found to be equivalent to the conventional fractionation, in terms of palliative benefits and response with about 75% incidence of grade III mucositis. Various dose fractionation regimens have been reported from major institutes of India, like Tata Memorial Hospital,[13] All India Institute of Medical Sciences (AIIMS),[14] and PGI Chandigarh.[56] The AIIMS study used a schedule of 20 Gy in 5 fractions over 1 week for 505 patients. Patients with more than 50% objective response received an additional dose escalation to 70 Gy (30% of 505 patients). Patients who received palliative radiotherapy alone without dose escalation had a 47%-59% symptom relief, median survival of 6.5 months, 1-year overall survival of 30%.[14] At Tata Memorial Hospital, Agarwal et al.,[13] used a schedule of 40 Gy in 16 fractions. Patients who had partial response had a dose escalation up to 50 Gy in 20 fractions. The incidence of grade III mucositis was 69%. About 74% patients had more than 50% symptom relief. Overall progression-free survival was 55% at 1 year. The cohort had 22% less than T4 disease, 17% less than N2 disease. Weight (>50 kg) and radiotherapy dose (>40 Gy) were significant prognostic factors for progression-free survival. Two palliative radiotherapy schedules were reported from PGI Chandigarh for a small group of patients (30 Gy in 10 fractions for n = 25 and Quad Shot regimen for n = 15). In the former study, all the 22 out of 25 patients had relief of pain which lasted for about 3 months. Only 16 patients came for follow-up and more than 50% patients had to travel over 50 km. to come to the hospital.[5] This is a real situation in India, and our experience parallels to that of the authors. In our schedule, about 73% patients completed the planned treatment without any interruption. The incidence of grade III or more mucositis was acceptable. More than 80% patients achieved palliation of presenting distressing symptoms. The protocol leads to significant reduction in hospital stay time for patients and machine time for treatment. The rate of hospitalization due to treatment was reasonably low. In our study, 88% patients had significant pain relief and only 18% patients had grade III mucositis. The median survival was 7 months which is comparable to that in the literature. The previous four studies from India and the present study clearly shows the relevance, benefit, and feasibility of hypofractionated short course radiotherapy in this condition.
CONCLUSIONS
This study shows the feasibility and benefits of hypofractionated palliative radiotherapy for inoperable head and neck cancer in terms of patient compliance, reduced side-effect, and effective palliation. There was significant improvement in presenting distressing symptoms and pain. Overall survival was comparable to similar other studies. This schedule would help to reduce the burden on the treatment machine and would be highly relevant in developing countries like India.
Source of Support: Nil.
Conflict of Interest: None declared.
REFERENCES
- Head and neck cancer in India: Need to formulate uniform national treatment guideline? Indian J Cancer. 2012;49:6-10.
- [Google Scholar]
- Survival from head and neck cancer in Mumbai (Bombay), India. Cancer. 2000;89:437-44.
- [Google Scholar]
- Burden of cancer and projections for 2016, Indian scenario: Gaps in the availability of radiotherapy treatment facilities. Asian Pac J Cancer Prev. 2008;9:671-7.
- [Google Scholar]
- Cancers of the head and neck region in developing countries. Radiother Oncol. 2003;67:1-2.
- [Google Scholar]
- Palliative radiotherapy in locally advanced head and neck cancer-A prospective trial. Indian J Palliat Care. 2004;10:19-23.
- [Google Scholar]
- Quad shot: A short but effective schedule for palliative radiation for head and neck carcinoma. Indian J Palliat Care. 2009;15:137-40.
- [Google Scholar]
- Palliative radiation therapy for head and neck cancer: Toward an optimal fractionation scheme. Head Neck. 2008;30:1586-91.
- [Google Scholar]
- Buccal mucosa cancer in South India. Etiologic and clinical aspects. Am J Roentgenol Radium Ther Nucl Med. 1966;96:6-14.
- [Google Scholar]
- Indian Council of Medical Research ND: Guidelines for management of buccal mucosa cancers. 2010.
- [Google Scholar]
- Radiation Therapy Oncology Group Acute radiation morbidity scoring criteria. Available from: http://www.rtog.org/ResearchAssociates/AdverseEventReporting/AcuteRadiationMorbidityScoringCriteria.aspx
- [Google Scholar]
- Palliative radiotherapy in head and neck cancers: Evidence based review. Indian J Palliat Care. 2006;12:44-50.
- [Google Scholar]
- A comparison of radiotherapy or radiochemotherapy with symptomatic treatment alone in patients with advanced head and neck carcinomas. Eur Arch Otorhinolaryngol. 2000;257:164-7.
- [Google Scholar]
- Hypofractionated, palliative radiotherapy for advanced head and neck cancer. Radiother Oncol. 2008;89:51-6.
- [Google Scholar]
- Short course palliative radiotherapy of 20 Gy in 5 fractions for advanced and incurable head and neck cancer: AIIMS study. Radiother Oncol. 2004;71:275-80.
- [Google Scholar]
- High fractional dose irradiation of advanced head and neck cancer. Implications for combined radiotherapy and surgery. Arch Otolaryngol. 1983;109:98-102.
- [Google Scholar]
- Hypofractionated radiotherapy for the palliation of advanced head and neck cancer in patients unsuitable for curative treatment–“Hypo Trial”. Radiother Oncol. 2007;85:456-62.
- [Google Scholar]
- Board of Faculty of Clinical Oncology, Royal College of Radiology. In: Radiotherapy Dose Fractionation. London: Royal College of Radiologists; 2006. p. :40-3.
- [Google Scholar]
- Squamous cell carcinomas metastatic to cervical lymph nodes from an unknown head and neck mucosal site treated with radiation therapy with palliative intent. Radiother Oncol. 2001;59:319-21.
- [Google Scholar]
- Outcome of treatment of 160 patients with squamous cell carcinoma of the neck staged N3a. Head Neck. 1990;12:483-7.
- [Google Scholar]
- Hypofractionated radiotherapy denoted as the “Christie scheme”: An effective means of palliating patients with head and neck cancers not suitable for curative treatment. Acta Oncol. 2009;48:562-70.
- [Google Scholar]
- The ‘QUAD SHOT’: A phase II study of palliative radiotherapy for incurable head and neck cancer. Radiother Oncol. 2005;77:137-42.
- [Google Scholar]
- Retrospective study of palliative radiotherapy in newly diagnosed head and neck carcinoma. Int J Radiat Oncol Biol Phys. 2011;81:958-63.
- [Google Scholar]
- Radiotherapy for head and neck cancer in elderly patients. Radiother Oncol. 2003;69:37-42.
- [Google Scholar]
- Comparative trial of large and small fractions in the radiotherapy of head and neck cancer. Clin Radiol. 1978;29:611-6.
- [Google Scholar]






