Translate this page into:
Quality of Life in Cancer Patients Undergoing Chemotherapy in a Tertiary Care Center in Malwa Region of Punjab
Address for correspondence: Dr. Harminder Singh; E-mail: dr_harminderchahal@rediffmail.com
This is an open-access article distributed under the terms of the Creative Commons Attribution-Noncommercial-Share Alike 3.0 Unported, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
This article was originally published by Medknow Publications & Media Pvt Ltd and was migrated to Scientific Scholar after the change of Publisher.
Abstract
Objective:
The objective of this prospective, non-interventional, 4-month observational study was to analyze and compare patient-reported quality of life (QOL) and their physical/psychosocial symptom burden during their respective chemotherapy sessions.
Materials and Methods:
A prospective and descriptive study was carried out jointly by Pharmacology and Oncology Departments of a tertiary care center in Malwa region of Punjab. The data collection was performed by administering validated questionnaire/response after taking informed consent.
Results:
A total of 131 cancer patients were recruited with the mean age of 49.05 ± 14.35 (SD (standard deviation)) years. As per the QOL scoring of Global Health Status (GHS) and four items of symptom scale, that is, insomnia, pain, appetite loss, and constipation, and financial difficulties attained a significance difference. GHS significantly improved in group three as compared to group one, indicating that the patient's overall health/QOL improved as the chemotherapy session progressed.
Conclusion:
Although QOL scoring system did not show significant improvement in all areas (except insomnia, pain, appetite loss, constipation, and financial difficulties) with reference to their respective chemotherapy cycles, but a judicious diagnosis with an appropriate treatment including chemotherapy may lessen the negative perception of cancer as a deadly and fatal disease in our rural population.
Keywords
Chemotherapy
European Organisation for Research and Treatment of Cancer Quality of Life Questionnaire-Core 36
quality of life
INTRODUCTION
Cancer prevalence in India is estimated to be around 2.0-2.5 million, with over 7-8 lakh new cases identified every year and data enumerated specifically among males: 46-122 per 100,000 population and among females: 57-135 per 100,000 population. Cancer deaths reported per year: 4-5 lakh. More than 70% of the cases report for diagnosis and treatment in the advanced stages of the disease, leading to a poor survival and high mortality rate.[1]
Nowadays, Punjab is acknowledged as cancer bowl of India with rising burden of cancer that leads to additional load of noncommunicable diseases. A survey was conducted by the Department of Health and Family Welfare, Government of Punjab in 2009 in which 7,738 cases of cancer were identified and out of which 245 were in Faridkot District of Punjab.[1]
As contrary to above accounts, a new disclosure by a nationally representative survey published in Lancet, revealed that the cumulative risk index (probability of death due to cancer) in Punjab is less than the national average and even lesser than most of the states in the country.[2] The State of Punjab has been in focus because of a mixture of reports emerging in media which especially focused on the Malwa region of the state. It shows divergence on prevalence and incidence of data regarding cancer patients.
Cancer chemotherapy in the 20th century has been dominated by the development of genotoxic drugs, initiated by the discovery of the anticancer properties of nitrogen mustard and the folic acid analogue aminopterin in the 1940s.[3] Chemotherapy has been recognized to decrease the incidence of both local and systemic recurrence and improve overall survival of patients.[4] Combination chemotherapy is usually preferred to outweigh risk-benefit ratios, however, long-term cancer chemotherapy have its own impact on cancer survivors including QOL, psychosocial issues, and physical symptoms and particularly the adverse effects of systemic adjuvant therapy (chemotherapy).[56]
QOL is subjective and patients own judgment in this respect is a major determinant, in a way it is described as a “quality of being”.[7] Cancer and its treatment has a substantial impact on mental and social health and in conclusion, on QOL of patients.[89] In this new era of cancer management, more emphasis is given on QOL rather than on quantity of life,; so in cancer patients where total cure is a remote attainment, measurement of QOL and follow-up may indicate acceptance, adaptation of disease, and chemotherapy.[101112]
Health-related QOL (HRQOL) measures (instruments and questionnaires) are well-defined questionnaires that gauge individuals' observation of their own physical, mental, and social health grade, or aspects of their health status resulting from cancer and its treatment.[13]
Many cancer-specific QOL measures have been developed, such as the Functional Adjustment to Cancer Therapy (FACT), European Organization for Research and Treatment of Cancer Quality of Life Questionnaire-C30 (EORTC QLQ-C33), Functional Living Index-Cancer (FLIC), and Cancer Rehabilitation Evaluation System (CaRES/CaRES-SF).[1415]
The objective of this prospective, non-interventional, 4-month observational study was to analyze and compare patient-reported QOL and their physical/psychosocial symptom burden, measured by completion of validated questionnaires in heterogeneous cancer cohort patients during their respective chemotherapy sessions.
MATERIAL AND METHODS
A total of 131 cancer patients were recruited in the present non-interventional, prospective clinical investigation analysis. The time duration was 4 months. The study was conducted by Oncology and Pharmacology Department of a tertiary care center in Malwa region of Punjab after taking permission from the Institutional Ethics Committee. Before the subjects were asked to participate and fill QOL questionnaire, a formal consent was obtained from all of them. The following inclusion criteria were outlined in advance before recruiting patients for study:
-
Diagnosed with cancer and visiting the institution to receive chemotherapy
-
No history of other chronic disease such as diabetes or heart disease
-
No known mental problem or being treated with psychotropic drugs.
After translating the EORTC QLQ-C30 in local language (Punjabi), standardized and modified questionnaires were used to measure QOL in the patients.
EORTC QLQ-C30 version 3
The EORTC QLQ-C30 version 3 questionnaire consists of five functional scales (physical, role, emotional, cognitive, and social), a scale for Global QOL, and nine symptom scales (fatigue, pain, nausea/vomiting, dyspnea, appetite loss, sleep disturbance, constipation, diarrhea, and financial difficulties). All measures are scaled from 0 to 100 with higher scores in symptom scales indicate a more severe problem (i.e. more severe symptoms).[1516]
We used a linear transformation to standardize the raw score, so that scores range from 0 to 100; a higher score represents a higher ("better") level of functioning GHS or a higher (“worse”) level of symptoms.[15] The patients were categorized as those with varied number of chemotherapy cycles as shown in Table 1.

Data collection
Baseline information included demographic data, for example, age, gender, education level, occupation, and marital status. Clinical information included type of cancer, family history, relapse, and duration and number of chemotherapy cycles.
Statistic
Baseline distinctiveness (demographic, clinical, and HRQOL) were summarized by descriptive statistics. Mean, percentages, and standard deviation (SD) were calculated wherever appropriate. HRQOL and clinical characteristics were compared among three distinct categories of participants [Table 1]. The comparison among three distinctive groups (varied number of chemotherapy cycles) was done by analysis of variance (ANOVA) with Tukey's honestly significant difference test (T-HSD) except in symptom score with skewed data; we applied nonparametric Kruskal-Wallis test. All P ≤ 0.05 were considered as significant.
RESULT
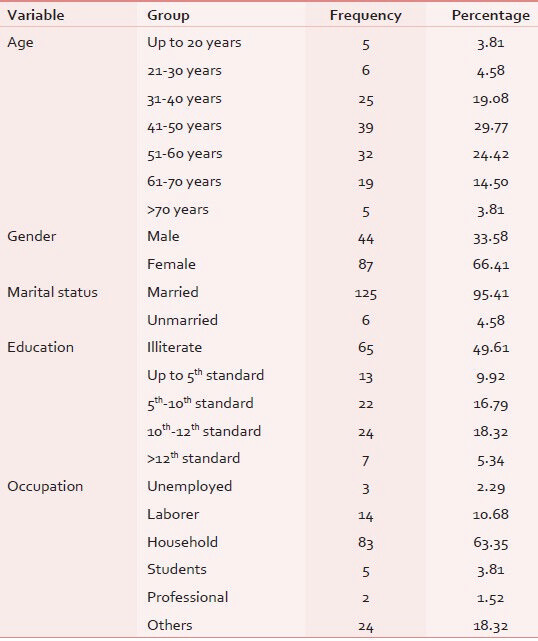
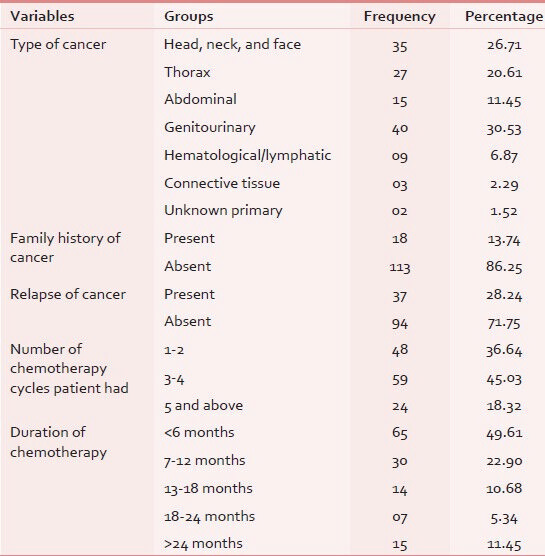
All 131 eligible patients participated in the study with response rate of 100%. The mean age of the participants was 49.05 years ± 14.35 (mean ± SD) with most common age group being 41-50 years with female preponderance (66%). Sixty-five patients (49.61%) were illiterate and majority were (65.64%) unemployed as shown in Table 2. Out of our 131 subjects, 30.53% were suffering from genitourinary carcinoma in which majority of cases were of cervix and endometrial cancers (11.45%), followed by ovary and testis (10.68%). Family history was seen only in 13.74%, in which no significant hereditary correlation was observed. Only 2.29% of patients had breast cancer relative (mother/grandmother) suffering in family. The cancer-specific variable tabulated in Table 3.
Mean and standard deviation of individual item in all three scales were calculated in all three groups and statistical inference was drawn using analysis of variances (ANOVAs) for any key significance with post ANOVA Tukey's HSD to establish intragroup characterizations as shown in Table 4.
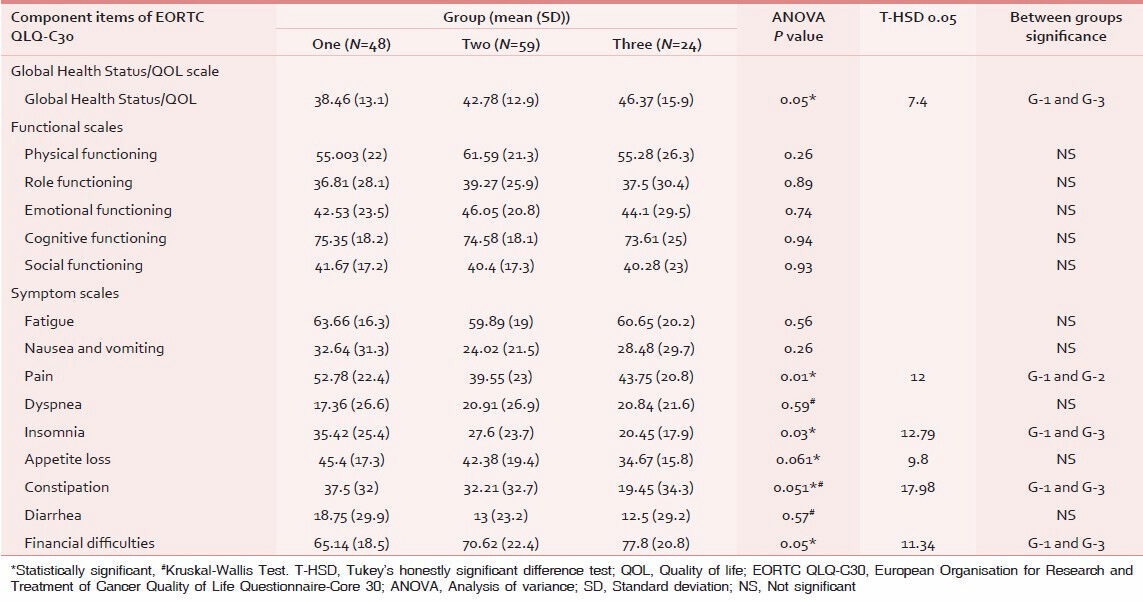
As per the QOL scoring, GHS and four items of symptom scale, that is, insomnia, pain, appetite loss, and financial difficulties attained a significant difference [Figures 1 and 2]. GHS significantly improved in group three as compared to the group one, indicating that the patient rated their overall health/QOL improved and better as the chemotherapy session progressed as shown in Figure 3. Statistical significant observed in pain parameter in G-1 and G-2, but surprisingly the improvement from G-2 to G-3 was not as expected. Five functional scales did not improve, rather remained invariable throughout the study [Figure 4]. A linear down regression was noticed in GHS with increasing age [Figure 5].
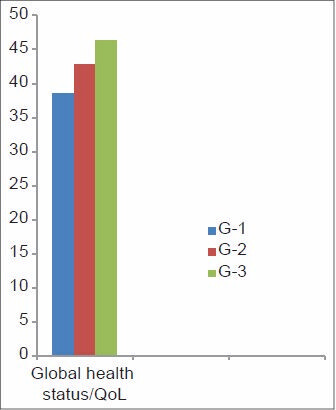
- Global health status/quality of life (QOL) scale scoring
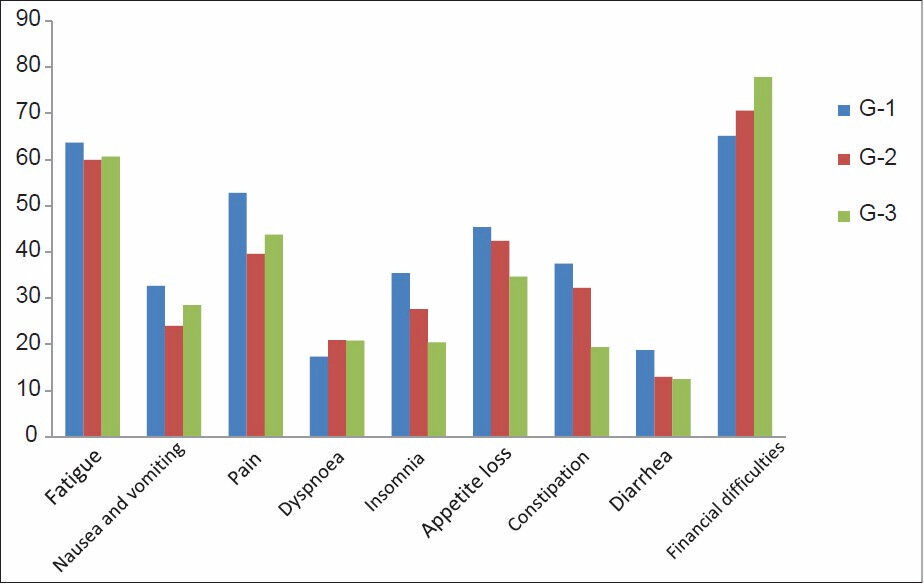
- Symptom scale scoring status
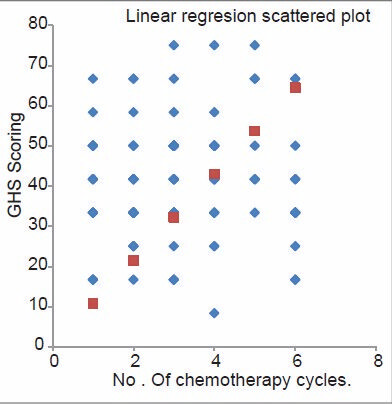
- Linear regression graphs of Global Health Status and number of chemotherapy cycle given
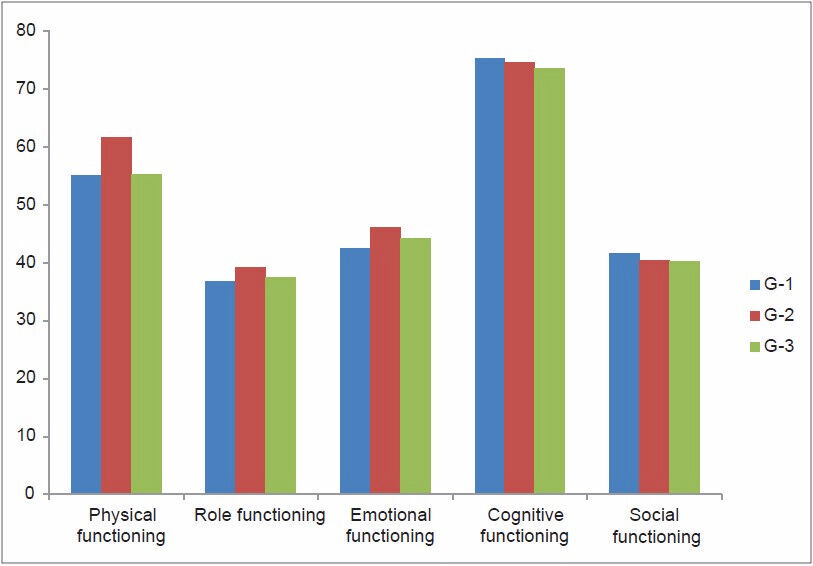
- Functional scale scoring status

- A linear regression graph of Global Health Status and age of patients.
DISCUSSION
In current medicine practice, the evaluation of a patient's health is based not only on clinical or laboratory indicator but also on holistic advances that includes the evaluation of the consequences of diagnosis or therapy/treatment, which perhaps indicate the progress of QOL in either way. So in cancer care, “global well-being” including physical, emotional, mental, social, and behavioral components is the main surrogate objective apart from concluding cure.
Nowadays, QOL has been introduced as an endpoint for treatment particularly in advanced cancer stages and it also is an early indicator of disease progression. A number of valid QOL tools have become accessible to determine HRQOL.[171819]
The most widely applicable instrument to measure the QOL in cancer patients is the EORTC QLQ-C30. Using this method, the current study assessed the QOL in cancer patients undergoing chemotherapy. Several studies support our findings on the influence of chemotherapy on QOL among the cancer patients.[202122]
The present study shows that improvement of QOL in cancer patient can be conceded by means of chemotherapy. However, this is not always the case. For example, Nemati et al., reported that the level of QOL in the patients with leukemia undergoing chemotherapy was 87.5% lower than that in the control group.[23] The differences might be due to different patients' population (sample size or patient age), cancer types, or may be due to wide-ranging toxicity of chemotherapy agents.
Statistical significant improvement was observed in pain parameter between group one and two, but surprisingly the improvement from group two to three was not as per expectation. This may be attributed to the subsequent adverse reactions that follow the advance stage of chemotherapy (like neuropathies or subjective variations). However, many other study quote amelioration of pain severity.[242526]
None of the functional scale items turned significant in our study including physical functioning, role functioning, emotional functioning, and cognitive and social functioning. Perhaps they remain stable during the progression of chemotherapy sessions with minor alteration which is in accordance with the study conducted by Zabernigg et al.[27]
Insomnia, appetite loss, and constipation significantly improved in majority of patients; in contrast, there was significant increase of financial burden due to high cost of drugs and loss of earning days during successive chemotherapy sessions.
According to the findings of the present study, statistically there was no significant correlation observed between demographic variables such as time passed from diagnosis, income level, marital status, and employment status with QOL; except younger age group who showed significant increase in GHS score and this has been shared by other studies.[2829] It should be noted that a considerable number of patients in the present study were illiterate or less educated; therefore, data collection in all the patients was assisted by medical students. So, it is likely that some questions are not precisely answered and it was one of the limitations of the study. Being heterogeneous groups (including all type of cancers, stages, etc.), without considering the stages and grading we encountered majority of patients at their variable stages of disease progression, so intragroup (within cancer) variations may be present.
Despite the limitations, the present study represents an attempt to understand the complicated interaction between cancer patients undergoing chemotherapy and their response to QOL domains. Our study is the first of its type which in depth assessed the chemotherapy response in terms of improvement in QOL in an area where the cancer incidence is alarmingly high, leading to panic alarm in rural population of Punjab.
CONCLUSION
A comprehensive assault by cancer surely disrupts the normal well-being of patients or we can say that cancer is an important health issue influencing QOL. The frequently considered efficacy criteria of cancer therapy are usually insufficient; if blended with individual's perspective (measured as HRQOL), than only it signifies a holistic approach toward the disease process. So well thought-out endpoint in cancer management with an assortment of conventional therapy and QOL measure will pronounce a better long-term goal. Education, awareness, and rehabilitation support are few minor but important things which need to be well-synchronized among government and social support organizations. A more comprehensive study is needed to establish the correlation of QOL with stage, grade, recurrence, metastasis of specific cancers, etc.
Source of Support: ICMR-STS-2013 Project.
Conflict of Interest: None declared.
REFERENCES
- Project Proposal September 2012. State Health Systems Resource Centre National Rural Health Mission. Department of Health and Family Welfare, Government of Punjab. Available from: pbhealth.gov.in/Final%20State%20Proposal.pdf
- [Google Scholar]
- Million Death Study Collaborators. Cancer mortality in India: A nationally representative survey. Lancet. 2012;79:1807-16.
- [Google Scholar]
- The use of nitrogen mustards in the palliative treatment of carcinoma. Cancer. 1948;1:634-56.
- [Google Scholar]
- Health professionals in the treatment of cancer and principles of cancer chemotherapy. In: Airley R, ed. Cancer Chemotherapy—Basic Science to the Clinic UK. Vancouver: Wiley-Blackwell; 2009.
- [Google Scholar]
- Fatigue in long-term breast carcinoma survivors: A longitudinal investigation. Cancer. 2006;106:751-8.
- [Google Scholar]
- Quality of life: A phenomenological perspective on explanation, prediction, and understanding in nursing science. ANS Adv Nurs Sci. 1985;8:1-14.
- [Google Scholar]
- Nursing Patients with Cancer: Principles and Practice (1st ed). Philadelphia, PA: Churchill Livingstone; 2006.
- Characteristics and quality of life analysis of caregivers of cancer patients. Med Oncol. 2010;27:607-17.
- [Google Scholar]
- QOL Researchers Have New Tool and New Focus on Measurement. J Natl Cancer Inst. 2008;20(100):234-6.
- [Google Scholar]
- Health-related Quality of life measurement. Int J Health Care Qual Assur. 2008;21:365-73.
- [Google Scholar]
- Quality of life data as prognostic indicators of survival in cancer patients: An overview of the literature from 1982 to 2008. Health Qual Life Outcomes. 2009;7:102.
- [Google Scholar]
- Assessing quality of life in adult cancer survivors (QLACS) Qual Life Res. 2005;14:1007-23.
- [Google Scholar]
- The European Organization for Research and Treatment of Cancer QLQ-C30: A quality-of-life instrument for use in international clinical trials in oncology. J Natl Cancer Inst. 1993;85:365-76.
- [Google Scholar]
- The EORTC QLQ-C30 Scoring Manual (3rd ed). Brussels: Brussels, EORTC Quality of Life Group; 2001. p. :86.
- Long-term Quality of Life after total laryngectomy and postoperative radiotherapy versus concurrent chemoradiotherapy for laryngeal preservation. Laryngoscope. 2008;118:300-6.
- [Google Scholar]
- European Organisation for Research and Treatment of Cancer (EORTC) Data Center and Quality of Life Group. Health related QOL outcomes in cancer clinical trials. Eur J Cancer. 2005;41:1697-709.
- [Google Scholar]
- The clinical value of Quality of Life assessment in oncology practice: A qualitative study of patient and physician views. Psychooncology. 2008;17:690-8.
- [Google Scholar]
- Linking clinical variables with health-related Quality of Life. A conceptual model of patient outcomes. JAMA. 1995;273:59-65.
- [Google Scholar]
- Quality of Life of 5-10 year breast cancer survivors diagnosed between age 40 and 49. Health Qual Life Outcomes. 2004;2:25.
- [Google Scholar]
- 1st congress in quality of life. Tehran, Iran: Book of Abstracts; 2003. p. :25. Quality of life in cancerous adolescences undergoing chemotherapy
- Quality of life during chemotherapy in non-small cell lung cancer patients. Acta Oncol. 1989;28:29-33.
- [Google Scholar]
- Abeloff's Clinical Oncology (3rd ed). Philadelphia, PA: Churchill Livingstone Elsevier; 2004. p. :715-30. Cancer pain Section one: Symptom Management and Palliative Care. Part II. Problems common to Cancer and its Therapy
- The EORTC QLQ LC13: A modular supplement to the EORTC Core QOL Questionnaire (QLQ-C30) for use in lung cancer clinical trials. EORTC Study Group on Quality of Life. Eur J Cancer. 1994;30:635-42.
- [Google Scholar]
- QOL across chemotherapy lines in patients with cancers of the pancreas and biliary tract. BMC Cancer. 2012;12:390.
- [Google Scholar]
- Hope and Quality of Life, two central issues for cancer patients: A theoretical analysis. Cancer Nurs. 1995;18:355-61.
- [Google Scholar]
- Physical, emotional and social dimension of QOL among breast cancer women under chemotherapy. IJNR. 2007;1:55-65.
- [Google Scholar]






